Varian Medical (NYSE:VAR) said it won 510(k) clearance from the FDA for its RapidPlan treatment planning software.
Oncology
How do you stop an aggressive breast cancer? Keep it from recycling
By Tom Ulrich
Guided Therapeutics widens distribution after FDA disappointment

Georgie-based Guided Therapeutics (OTC:GTHP) hasn’t won many points after announcing increased distribution for its LuViva advanced cervical scanning system.
Delcath lays off 33% of its employees
Preventive mastectomy may not do much good, study says | MassDevice.com On Call

MASSDEVICE ON CALL — Double mastectomy rates have doubled over the last 10 years, including some women who’ve opted to remove a healthy breast after discovering early-stage breast cancer in the other, but the extreme measure may not be doing them much good.
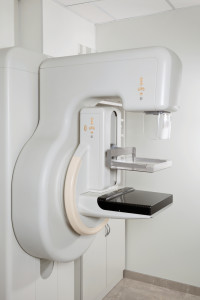
Breast cancer: Hologic touts ‘excellent’ results from 7-year MammoSite registry

Langer brainchild Bind Therapeutics launches $70.5M IPO

Serial entrepreneur and Massachusetts biotech super-star Dr. Robert Langer’s Bind Therapeutics last week launched an initial public offering aimed at raising $70.5 million from shares sold at $15 apiece.
The clinical-stage company, co-founded by Langer and Dr. Omid Farokhzad, is developing nanotech-based solutions for "programmable" therapeutics, starting a pair of cancer drugs.
American Cancer Society CMO: Money-driven hospitals push for dangerous tests

Routine screening has been touted as the most effective way to stem cancer deaths, but it may instead put patients in harm’s way, according to American Cancer Society chief medical officer Dr. Otis Brawley. Speaking at a seminar last week, Brawley warned that hospitals recommending regular screening may be looking out for their profits rather than their patients.
Delcath dives again on final FDA denial of liver cancer treatment
Elekta lands $25M Gamma Knife order from China

Implantable melanoma vaccine proceeds to human testing | MassDevice.com On Call

MASSDEVICE ON CALL — After a quick FDA turnaround, an implantable melanoma vaccine is beginning a Phase I clinical trial. The technology was co-developed by the Wyss Institute and Dana Farber Cancer Institute
Researchers developed a tiny, biodegradable disk made of growth factors and elements of each patient’s tumor. Once implanted, the disk dissolves and prompts the body to attack the tumor as though it were an infection.


