
Edwards Lifesciences
MassDevice.com +3 | The top 3 med-tech stories for March 26, 2012.

Say hello to MassDevice +3, a bite-sized view of the top three med-tech stories of the day. This feature of MassDevice.com’s coverage highlights our 3 biggest and most influential stories from the day’s news to make sure you’re up to date on the headlines that continue to shape the medical device industry.
If you read nothing else today, make sure you’re still in the know with MassDevice +3.
ACC 2012: Medtronic and Edwards rise on lower stroke rates in new TAVI data

ACC 2012: TAVI stands up to surgery at 2 years, but valve leak risk increases

Report: Medtronic to shutter Ventor’s Israeli shop | Wall Street Beat

Medtronic (NYSE:MDT) is shutting down heart valve maker Ventor Technologies, which it acquired in 2009, and moving its R&D efforts to other site, according to an Israeli business journal.
Netanya, Israel-based Ventor is developing an aortic replacement valve called the Engager. Medtronic bought the company for $325 million 3 years ago.
Analyst: CRM market “bad as it’s ever been” | Wall Street Beat

The cardiac rhythm management market is "about as bad as it has ever been," according to an analyst with Gabelli & Co., due in part to headwinds from a federal probe and a negative journal article.
FDA wrestles with mobile medical apps and self-screening devices | MassDevice.com On Call

MASSDEVICE ON CALL — The FDA will meet this month to consider the role the agency will play in mobile medical technologies and patient-driven screening and diagnostics.
The agency forecasts that patients will soon have access to technologies that allow them to self-screen for diseases or conditions via stand-alone kiosks or wireless diagnostic tools like blood pressure readers and blood glucose readers.
Cameron Health’s FDA date is good news for Boston Scientific

Cameron Health landed an April date with the FDA’s circulatory devices panel to review a pre-market approval application for its lead-free subcutaneous implantable cardioverter defibrillator, or S-ICD.
The S-ICD system, unlike traditional ICDs, sits just beneath the skin and does not rely on wires relayed through blood vessels and connected to the heart in order to provide heart rhythm therapy.
VCs press Senate on “IPO On Ramp” bill | Wall Street Beat

The National Venture Capital Assn. is spearheading a drive to get a bill through the U.S. Senate that would make it easier for small companies to raise money by going public.
The "IPO On Ramp" measure would ease the regulatory requirements for IPOs and temporarily reduce the reporting burdens imposed by the Sarbanes-Oxley Act.
Medtronic fires new salvo in ongoing aortic valve patent war with Edwards
Edwards warns on trial limits, doc licenses in Medicare’s TAVI reimbursement guidelines
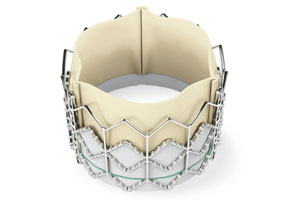
Edwards Lifesciences (NYSE:EW) had words of praise and caution about a proposed reimbursement scheme for its Sapien transcatheter aortic valve implant unveiled by the Centers for Medicare & Medicaid Services last month.
Edwards, which boasts the only TAVI device cleared for the U.S. market, asked Medicare to reconsider limiting future clinical trials and asked the agency to re-think proposed rules for procedure credentialing focused on individual physicians.



