
Boston Scientific’s (NYSE:BSX) lead-free S-ICD cardiac rhythm management made another strong showing, with results from an analysis of more than 880 patients showing relatively few complications.
The Medical Device Business Journal — Medical Device News & Articles | MassDevice

Boston Scientific’s (NYSE:BSX) lead-free S-ICD cardiac rhythm management made another strong showing, with results from an analysis of more than 880 patients showing relatively few complications.

Boston Scientific (NYSE:BSX) touted the results of a comparative analysis of patients treated with the subcutaneous implantable defibrillator, reporting that its lead-free ICD may be safer for patients.

Frequent medtech challenger Dr. Robert Hauser offered some words of warning for physicians clamoring over Boston Scientific’s (NYSE:BSX) wire-free implantable defibrillator: curb your enthusiasm.

A new study of the so-called “leadless” defibrillator made by Boston Scientific (NYSE:BSX) shows that the device matches the safety and performance of standard defibrillators, which use leads threaded through blood vessels to deliver shocks to the heart.
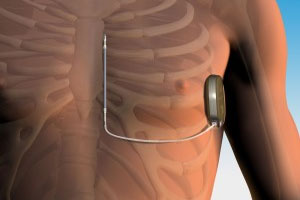
Boston Scientific (NYSE:BSX) and newly acquired Cameron Health won FDA approval for the world’s only commercially available lead-free implantable cardioverter defibrillator, the S-ICD.
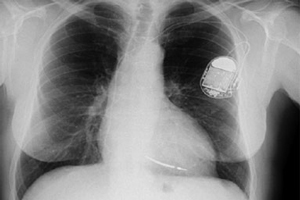
Innovative heart rhythm technologies that do away with the leads that "tether" the device to the heart may be the next big thing for heart failure patients, but the technology has a way to go before disrupting the market, according to panelists at the Cardiostim 2012 conference in France this week.
Getting rid of the leads that weave through the body to deliver shocks to the heart would mean doing away with risks including lead fractures, erosion and infections, Cleveland Clinic’s Dr. Bruce Wilkoff told an audience at a popular presentation.

Say hello to MassDevice +3, a bite-sized view of the top three med-tech stories of the day. This feature of MassDevice.com’s coverage highlights our 3 biggest and most influential stories from the day’s news to make sure you’re up to date on the headlines that continue to shape the medical device industry.
If you read nothing else today, make sure you’re still in the know with MassDevice +3.

Cameron Health’s under-the-skin defibrillator may take longer to respond to cardiac events than standard implantable cardiac defibrillators, but speed isn’t always the most important factor in saving a life.
Although Cameron’s S-ICD took longer than 18 seconds to respond in 12% of patients in the ongoing Effortless study, it always responded in time with life-saving therapy and did so while delivering fewer inappropriate shocks to the heart.

Boston Scientific (NYSE:BSX) and Cameron Health sealed the deal on a merger worth up to $1.35 billion.

