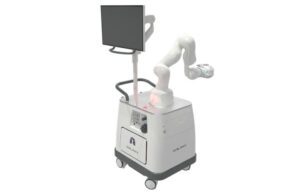
San Carlos, California-based Noah Medical received FDA clearance for the Galaxy system in March. The system provides bronchoscopic visualization and access. These capabilities provide diagnostic and therapeutic procedures in patient airways.
Galaxy features advanced imaging technologies that provide real-time location updates for potentially cancerous lesions. Noah Medical designed the technology to improve tool-in-lesion and diagnostic yield.
Dr. Kyle Hogarth conducted the first U.S. procedure at the University of Chicago Medicine.
“The Galaxy System is the first robot for peripheral lung navigation that includes integrated imaging, which is the solution bronchoscopists have been asking for,” said Hogarth. “We are excited to be the first hospital in the United States to implement this next-generation robotic solution for our patients which we believe will improve and expedite the early diagnosis and treatment of lung cancer.”
It’s the latest in a string of positive steps for Noah Medical, which raised $150 million a month after garnering FDA clearance. Late last month, the company also shared positive accuracy results from a study of its Galaxy system.
Noah Medical also said its first-in-human trial in Australia produced “overwhelmingly positive” physician feedback. Galaxy offers the company’s TiLT Technology with integrated tomosynthesis and augmented fluoroscopy. Its single-use disposable bronchoscope and small, compact footprint also enables easy integration into most bronchoscopy suites.
“We designed the Galaxy System in collaboration with physicians to provide unique clinical value to patients and improved procedural efficiency,” said Jian Zhang, Noah Medical founder and CEO. “We are thrilled to introduce the platform at UChicago Medicine, a pioneer in using life-saving technologies to fight lung cancer.”

