
A long-awaited merger between Johnson & Johnson (NYSE:JNJ) and Synthes Inc. may come to a close this week, after U.S. anti-trust regulators approved the $21.3 billion acquisition.
The Medical Device Business Journal — Medical Device News & Articles | MassDevice
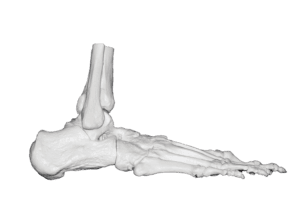 Welcome to MassDevice's Orthopedics Hub, your resource for the latest in orthopedic news. Stay informed about the innovations and business news shaping the ortho device industry.
Welcome to MassDevice's Orthopedics Hub, your resource for the latest in orthopedic news. Stay informed about the innovations and business news shaping the ortho device industry.
🦴 Narrow your focus with these categories:
Implants | Metal-on-Metal | Spine
Here are the top orthopedic device features from MassDevice and its sister publications (and scroll down to read the latest cardiology device news from MassDevice):
+ The biggest stories from AAOS 2024
+ The 10 largest orthopedic device companies in the world
+ The ZimVie Tether helps kids with scoliosis — if they can get it in time
+ Could 2023 be a boom year for orthopedic devices?
+ The top orthopedic device news out of AAOS 2023
🎙️ Tune in to the Ortho Innovation Talks podcast on the DeviceTalks podcast network.

A long-awaited merger between Johnson & Johnson (NYSE:JNJ) and Synthes Inc. may come to a close this week, after U.S. anti-trust regulators approved the $21.3 billion acquisition.

Orthopedics devices maker ConforMIS launched a $68 million funding round that will close at the end of this month.
ConforMIS is offering equity, securities and options with no minimum investor, according to regulatory filings.

Smith & Nephew (FTSE:SN, NYSE:SNN) shares dropped nearly 3% in London and opened down 2.8% on Wall Street after the company said it’s voluntarily yanking a component from 1 of its metal-on-metal hip implants.

MASSDEVICE ON CALL — A pair of important medical device industry legislations were momentarily combined when Sen. Orrin Hatch (R-Utah) announced plans to include language repealing the 2.3% medical device tax in a bill reauthorizing the user fees device and drug makers pay for FDA review.
The effort was short-lived. On Monday Sen. Dan Coates, a co-sponsor of the Hatch amendment, conceded that Hatch planned to find "another vehicle" for his tax repeal efforts so as to avoid stalling the FDA user fee agreements.

Medtronic’s (NYSE:MDT) bellwether 4th quarter and full-year earnings noted a continued decline in U.S. cardiac rhythm management and spinal products, but the slump may be slowing, the company said.

The U.S. Justice Dept. closed the books on a years-long investigation into Medtronic‘s (NYSE:MDT) controversial Infuse bone growth protein, the company announced today.
The office of the U.S. Attorney for the District of Massachusetts and the DoJ abandoned their civil and criminal probe into allegations of off-label marketing and concealed safety risks without finding evidence of wrongdoing, according to a press release.

