
On Feb. 8, The Food & Drug Administration unveiled a new, fast-track protocol for breakthrough medical technologies this week, saying its new “Innovation Pathway” aims to speed high-tech devices to market.
The Medical Device Business Journal — Medical Device News & Articles | MassDevice

On Feb. 8, The Food & Drug Administration unveiled a new, fast-track protocol for breakthrough medical technologies this week, saying its new “Innovation Pathway” aims to speed high-tech devices to market.

Aushon BioSystems Inc. partnered up with Peking University Cancer Hospital to develop colorectal cancer-detection technology, aiming to predict when colorectal cancer migrates to the liver.
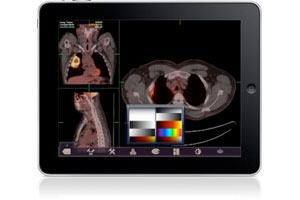
It took nearly three years and three 510(k) applications, but MIM Software finally won Food & Drug Administration clearance for its Mobile MIM app, the first-ever mobile radiology app to receive the agency’s seal of approval.
The process was so fraught with confusion and miscommunication — or worse, no communication — that MIM CTO Mark Cain likened the experience to falling into a "black hole."

Highlights of the important and interesting in the world of healthcare.
MASSDEVICE ON CALL — Study: Fetal surgery treats spina bifida. Surgery inside the womb to repair an unborn baby’s myelomeningocele, the most common form of spina bifida, "may result in better neurologic function than repair deferred until after delivery," according to a study published in The New England Journal of Medicine.

MassDevice keeps a close eye on public medical device companies, tracking their quarterly sales and earnings reports. For the most recent filings, check out our Earnings Roundup, where we collect each quarter’s reports.
Here’s a quick rundown of a few releases over the past couple day:

The internet can be a swamp of hackers, crackers, and hucksters attacking your systems for fun, profit and fraud. Defending your data and applications against this onslaught is a cold war, requiring constant escalation of new techniques against an ever increasing offense.
By Chris Seper

It’s hard to gauge the impact of The New York Times’ critique of WebMD. Is it just one columnist’s opinion? Or is it a signal that standards for online health advice are going to change?
Times columnist Virginia Heffernan cracked WebMD as “synonymous with Big Pharma Shilling” and the ultimate place for “hypochondria time suck.”

Contract research organization Boston Biomedical Associates will now have the added staff support of Creganna-Tactx Medical.
The two companies formed a joint venture under which Creganna-Tactx customers will have access to BBA’s clinical, regulatory and quality consulting services.
LifeBot develops tablet for prehospital patient care: LifeBot LLC implemented the HP Slate 500 as an in-ambulance computer and universal communication device. The tablet runs Windows 7 and can transmit live patient data to the receiving hospital via the DREAMS software, which was developed with U.S. Army Materiel Command, Texas A&M and UTHealth Science Center at Houston, according to the company.

Here’s a roundup of companies announcing clearances and approvals from the Food & Drug Administration and foreign regulatory agencies. Check out MassDevice’s compilation of the latest regulatory news for additional clearances and approvals.

