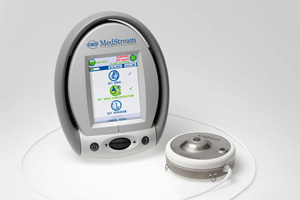
The FDA granted PMA supplemental approval to Codman & Shurtleff for the Medstream programmable infusion device, an implantable infusion pump and catheter used in treatment of movement disorders.
The Medstream system delivers continuous doses of an anti-spasm drug directly to he spinal canal to relieve spasticity, a disorder affecting more than 12 million people worldwide, which is often caused by stroke, cerebral palsy, multiple sclerosis or spinal cord injury, according to the company.