

The Medical Device Business Journal — Medical Device News & Articles | MassDevice



MASSDEVICE ON CALL — The U.S. Government accused 2 Florida radiation oncologists and a group of treatment centers of defrauding state and government health programs for more than $3.5 million by conducting unnecessary procedures and lying about them.
Since at least December 2007 and continuing through October 2011 the defendants allegedly billed Medicare and other health services for "unreasonable, unnecessary, or medically improper radiation diagnosis treatments provided to patients without complying with the regulations, protocols, and requirements for reimbursement."

MASSDEVICE ON CALL — Rep. Ed Markey (D-Mass.) launched a new bill aimed at strengthening the FDA’s oversight of medical devices.
The Safety of Untested & New Devices Act would give the FDA the power to request that companies submitting new devices speak for any recalls or safety issues in predicate devices.

President Barack Obama isn’t planning anything special to mark the anniversary of his signature health care reform bill, the Patient Protection & Affordable Care Act, which he signed into law 2 years ago this month.
There will be no public remarks or special press conferences to defend the measure this week as the case inches closer to opening arguments in the Supreme Court, administration officials said.

MASSDEVICE ON CALL — The 2012 list of the world’s most ethical companies included a handful of medical device makers and group purchasing organizations, according to the research-based Ethisphere Institute.

MASSDEVICE ON CALL — A team of U.K. researchers is calling for a ban on metal-on-metal hip implants, citing studies that demonstrate that they fail more often than other artificial hips, especially when used in women.

MASSDEVICE ON CALL — Brain stimulation using electrical currents is a safe and effective depression therapy and may even have other surprise perks for the body, according to research conducted at the University of New South Wales.
More than half of all patients in what researchers called the largest and most definitive study of transcranial direct current stimulation experienced "substantial improvements" after getting the therapy, according to a press release.

MASSDEVICE ON CALL — Medistem Inc. (PINK:MEDS) partnered with Yale University on diabetes treatment using stem cells to generate pancreatic islets from endometrial regenerative cells.
Pancreatic islets effectively treated diabetes in animal models, and Medistem is also studying ERCs as therapy for critical limb ischemia and congestive heart failure, both complications of uncontrolled diabetes.
Updated March 7, 2012, at 1:30 p.m. with comments from GE Healthcare.

MASSDEVICE ON CALL — GE Healthcare (NYSE: GE) announced another round of layoffs at its South Burlington, Vt., facility, a move it said is a result of declining demand for health care IT and performance solutions businesses.
The move is the "subsequent action" of layoffs numbering around 50 workers at the same facility last month, GE spokesman Corey Miller told MassDevice.
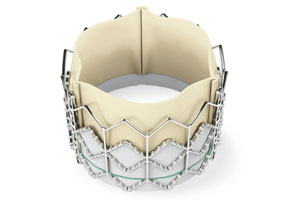
Edwards Lifesciences (NYSE:EW) had words of praise and caution about a proposed reimbursement scheme for its Sapien transcatheter aortic valve implant unveiled by the Centers for Medicare & Medicaid Services last month.
Edwards, which boasts the only TAVI device cleared for the U.S. market, asked Medicare to reconsider limiting future clinical trials and asked the agency to re-think proposed rules for procedure credentialing focused on individual physicians.

