Food & Drug Administration (FDA)
FDA OKs SynCardia trial for bridge-to-transplant indication
SynCardia Systems said today that it won FDA approval for a clinical trial of its artificial heart as a bridge to transplant for patients with heart failure.
TAVI: FDA adds ‘valve-in-valve’ indication for Medtronic’s CoreValve
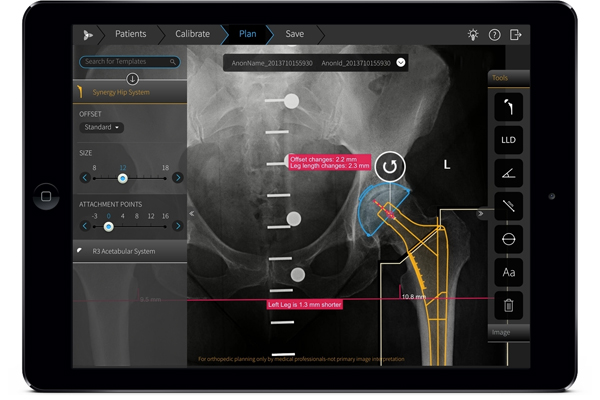
FDA green-lights Brainlab’s TraumaCad Mobile app
In swan song, FDA chief Hamburg defends approval process
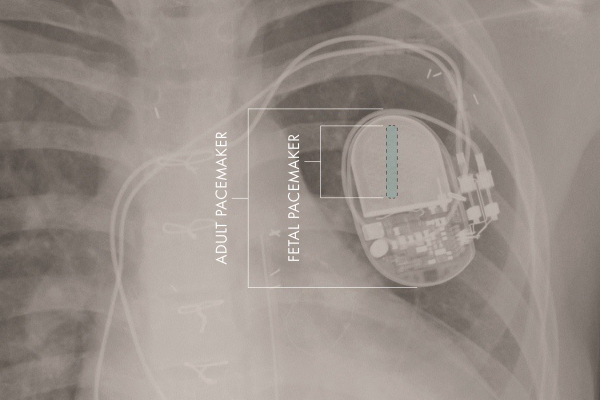
FDA grants humanitarian use device status to fetal micro-pacemaker
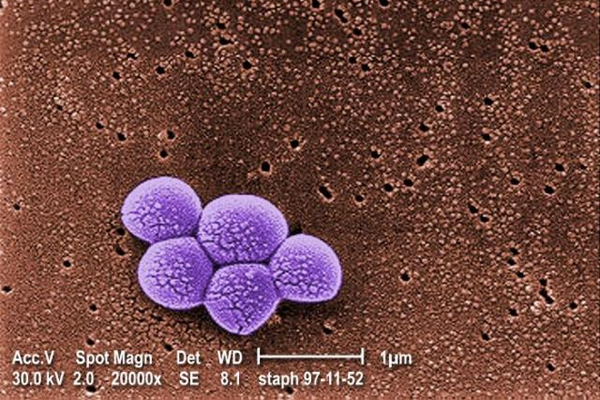
White House tackles superbugs
(Reuters) – The White House is due to issue an ambitious plan to slow the growing and deadly problem of antibiotic resistance over the next 5 years, 1 that requires massive investments and policy changes from a broad array of U.S. government health agencies, according to a copy of the report reviewed by Reuters.