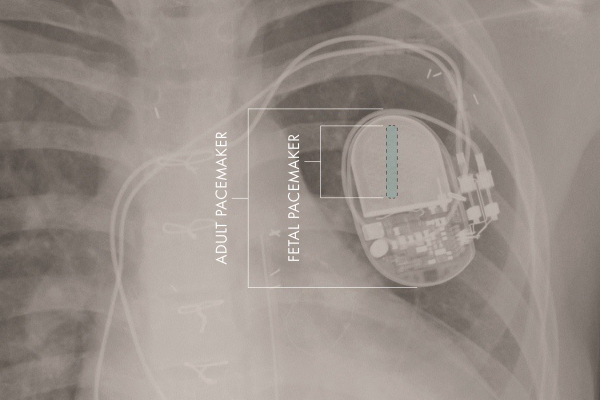
Researchers at Children’s Hospital Los Angeles and the University of Southern California said the FDA granted humanitarian use device status to the fetal micro-pacemaker they designed.
Humanitarian Device Exemption (HDE)
FDA approves Abiomed’s Impella RP
Medtronic wins HDE for Enterra II neurostim device
Hypertension: CVRx wins FDA nod for Barostim neo legacy
XVIVO Perfusion wins FDA approval for donor lung transport system

The FDA granted Humanitarian Use Device approval for Sweden-based XVIVO Perfusion for its XPS tissue transport system for preserving donor lungs.
Press Release: FDA approves donor lung preservation device that may result in more lung transplants

FDA updates medtech fast-track review for rare treatments | MassDevice.com On Call

MASSDEVICE ON CALL — U.S. healthcare regulators released new documents in efforts to clarify the FDA’s Humanitarian Device Exemption pathway, a less-stringent review process for medical devices that treat rare diseases.
The FDA issued a new draft guidance that would allow devices to bypass some of the required safety and efficacy hurdles if it is intended to treat a population of less than 4,000 patients.
FDA sets a date to review XVIVO’s lung perfusion system

Sweden-based XVIVO Perfusion landed a date with the FDA to review Humanitarian Device Exemption for its XVIVO Perfusion System for preserving donor lungs.
XVIVO will meet on March 20 with the FDA’s Gastroenterology & Urology Devices Panel to review data on the device, which provides continuous perfusion of otherwise unacceptable lung tissue, allowing clinicians to reevaluate the organs for transplantation.
Invivo starts first human trial for spinal cord scaffold

InVivo Therapeutics (OTC:NVIV) said the FDA has given the company the green light to begin the first human clinical studies of its bioploymer scaffold, which treats traumatic spinal cord injury.
The Cambridge, Mass.-based company said it is gearing up for safety studies in 5 patients over 15 months, and is shooting for Institutional Review Board (IRB) approval at Massachusetts General Hospital.
InVivo Therapeutics’ spinal cord scaffold wins FDA nod

InVivo Therapeutics (OTC:NVIV) said the FDA deemed its biopolymer scaffold, designed to treat spinal cord injuries, to be a humanitarian use device.
The designation means the InVivo scaffold can seek expedited approval from the FDA for a pivotal trial of the device. HUD-based exemptions cover devices aimed at treating rare diseases and health problems, so-called "orphan" conditions.
Syncardia nabs 2 Humanitarian Use Device wins for its smaller artificial heart
SynCardia Systems won 2 new Humanitarian Use Device designations for its 50cc Total Artificial Heart as a cardiac bridge transplant device.
The new HUD designations apply to use with children, smaller women and other people with small stature. The Tuscon, Ariz.-based device manufacturer designed this device as a smaller version of its 70cc temporary Total Artificial Heart, approved in 2004, according to a press release.