(Reuters) — In her final speech after 6 years as commissioner of the FDA, Dr. Margaret Hamburg defended the agency’s approval standards against critics seeking weaker regulatory standards.
Food & Drug Administration (FDA)
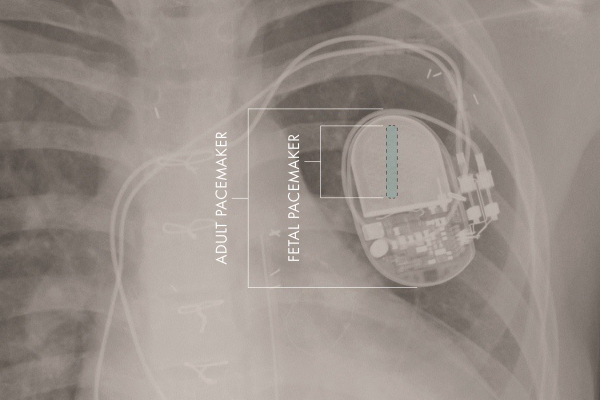
FDA grants humanitarian use device status to fetal micro-pacemaker
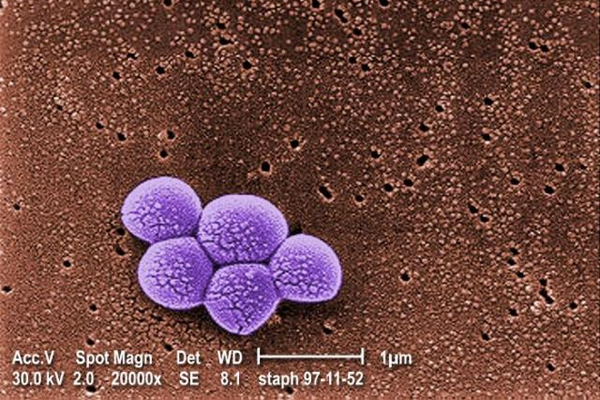
White House tackles superbugs
(Reuters) – The White House is due to issue an ambitious plan to slow the growing and deadly problem of antibiotic resistance over the next 5 years, 1 that requires massive investments and policy changes from a broad array of U.S. government health agencies, according to a copy of the report reviewed by Reuters.
FDA clears Stryker’s Lite lumbar plating system
Olympus issues ‘urgent’ steps for cleaning device linked to superbugs
CRM: BSX’s gain is STJ’s loss | Medtech Wall Street news for the week of March 23, 2015
Wright Medical wins FDA nod for Salvation external fixation device
UPDATE: Abiomed soars to new all-time high on FDA nod
BSX wins CE Mark, FDA nod for new S-ICD | Regulatory news for the week of March 23, 2015
FDA’s draft guidance on medical device accessories: Potential considerations for manufacturers
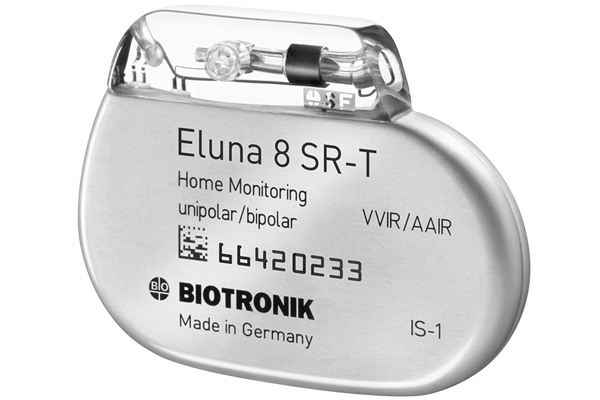
FDA approves Biotronik’s Eluna MRI-safe pacemaker
Biotronik said today that the FDA approved its ProMRI Eluna MRI-safe pacemaker, making it the only company with single- and dual-chamber MRI-safe pacemakers on the U.S. market.