
St. Jude Medical (NYSE:STJ) said it won CE Mark approval in the European Union for the 25mm version of its Portico replacement heart valve.
The Medical Device Business Journal — Medical Device News & Articles | MassDevice
 Welcome to MassDevice's Cardiovascular Hub, your resource for the latest news in cardiac implants, structural heart, heart valves, stents and more. Stay informed about the innovations and business news shaping the cardiovascular device industry.
Welcome to MassDevice's Cardiovascular Hub, your resource for the latest news in cardiac implants, structural heart, heart valves, stents and more. Stay informed about the innovations and business news shaping the cardiovascular device industry.
🫀 Narrow your focus with these categories:
Cardiac Implants | Structural Heart | Pulsed-Field Ablation
Here are top cardiology features from MassDevice and its sister publications (and scroll down to read the latest cardiology device news from MassDevice):
+ LVADs save lives: So why aren’t more available?
+ How Medtronic uses nitinol to improve the structure and effectiveness of heart devices
+ How Abbott designed the world’s first dual-chamber leadless pacemaker system
+ J&J used RWE for expanded indications — and you can, too
+ 7 cardiology startups you need to know

St. Jude Medical (NYSE:STJ) said it won CE Mark approval in the European Union for the 25mm version of its Portico replacement heart valve.
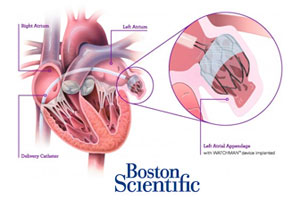
The FDA’s Circulatory Systems Devices Panel today voted 13-1 to recommend approval of Boston Scientific’s(NYSE:BSX) Watchman 1st-of-its-kind left atrial appendage (LAA) closure device for reducing the risk of stroke and systemic embolism.

A conservative forecast for 2014 sent shares of Edwards Lifesciences (NYSE:EW) down more than 5% yesterday and kept them flat today, as the medical device company said it expects more competition next year for its flagship Sapien replacement heart valve.

Bolton Medical launched phase II of its U.S. clinical trial for its Treovance abdominal stent graft system and corresponding Navitel delivery system.
The Treovance stent graft is designed to be adaptable and flexible in order to accommodate a wide range of anatomies. The Navitel delivery system includes a proximal clasping system for precise delivery.
The phase II study will treat 150 patients with infrarenal aortic aneurysms, enrolled at 30 institutions located throughout the U.S., according to the press release.

Boston Scientific (NYSE:BSX) may have to confront some tough questions when it faces a panel of FDA experts reviewing its Watchman stroke-preventing heart implant.

Boston Scientific (NYSE:BSX) said it inked a deal with The Medicines Co. to promote its Promus Premier coronary stent in the U.S.

Claret Medical said it filed asked for an investigative device exemption from the FDA for a clinical trial of its Sentinel cerebral protection system.
Santa Rosa, Calif.-based Claret wants to conduct a multicenter study using the Sentinel for embolic protection during transcatheter aortic valve implantation, comparing use of the Sentinel device with standard care.

Emerging medical device maker Transcatheter Technologies GmbH unveiled an early look at clinical results for its 3rd-generation Trinity transcatheter aortic valve implantation platform, which the company calls "the world’s 1st ‘truly repositionable’ and, therefore, best TAVI system."
Palmaz Scientific is nearing the close of its $30 million funding bid, with just $3.7 million needed to close the round, according to regulatory filings.
The company revealed in an SEC document that it raised $26.3 million from 261 unnamed investors, with as much as 10% of the take going to cash commissions for its placement agents.

Sorin Group (BIT:SRN) said it won CE Mark approval in the European Union for the XL version of its Perceval sutureless aortic valve.
Milan-based Sorin said the new approval is for treating patients with annulus sizes ranging from 19 mm to 27 mm using the Perceval device.

