
Edwards Lifesciences (NYSE:EW) said it won an investigational device exemption from the FDA to run a clinical trial of its Sapien 3 replacement heart valve in intermediate-risk patients.
The Medical Device Business Journal — Medical Device News & Articles | MassDevice
 Welcome to MassDevice's Cardiovascular Hub, your resource for the latest news in cardiac implants, structural heart, heart valves, stents and more. Stay informed about the innovations and business news shaping the cardiovascular device industry.
Welcome to MassDevice's Cardiovascular Hub, your resource for the latest news in cardiac implants, structural heart, heart valves, stents and more. Stay informed about the innovations and business news shaping the cardiovascular device industry.
🫀 Narrow your focus with these categories:
Cardiac Implants | Structural Heart | Pulsed-Field Ablation
Here are top cardiology features from MassDevice and its sister publications (and scroll down to read the latest cardiology device news from MassDevice):
+ LVADs save lives: So why aren’t more available?
+ How Medtronic uses nitinol to improve the structure and effectiveness of heart devices
+ How Abbott designed the world’s first dual-chamber leadless pacemaker system
+ J&J used RWE for expanded indications — and you can, too
+ 7 cardiology startups you need to know

Edwards Lifesciences (NYSE:EW) said it won an investigational device exemption from the FDA to run a clinical trial of its Sapien 3 replacement heart valve in intermediate-risk patients.

California medtech company Direct Flow Medical notched some major milestones today, announcing a European regulatory win and completed enrollment in a U.S. feasibility trial for its transcatheter aortic valve implantation systems.

Boston Scientific (NYSE:BSX) today announced the appointment of Dr. Craig Thompson as senior vice president and chief medical officer for the medtech giant’s interventional cardiology business.

Paris-based medical device maker Carmat this month successfully implanted its 1st bioprosthetic artificial heart, according to a company statement.
The company conducted the implant as part of a feasibility study, in accord with approval studies with the French health authority, the ANSM.

Xlumena won FDA approval to market its Axios stent and delivery system, landing approval through the FDA’s less-stringent de novo review pathway.
The stent is the 1st specifically designed for the drainage of a pancreatic pseudocyst by creating a new, temporary opening between the pancreas and the gastrointestinal tract.
Patients getting drug-eluting stents may be able to cut their post-procedure blood thinner regimen by as much as 75%, according to study published this week in the Journal of the American Medical Assn.

Avinger said it closed its latest significant funding round, where both new as well as existing investors participated in the financing.
The Redwood City, Calif.-based company brought in an unspecified amount of money, saying that the funds are expected to go towards continuing the company’s operations.
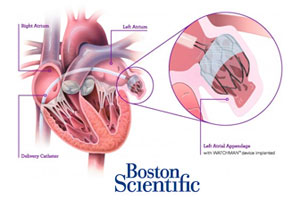
The FDA’s Circulatory Systems Devices Panel voted 13-1 yesterday to recommend approval of Boston Scientific‘s (NYSE:BSX) Watchman heart implant for reducing the risk of stroke and systemic embolism.

Cook Medical said it started a multi-center clinical trial of its Evolution esophageal stent.
The study is designed to test the possibility of removing the self-expanding, covered metal stent after treatment of malignant and benign strictures including strictures, fistulas or perforations.

