The FDA’s Circulatory Systems Devices Panel today voted 13-1 to recommend approval of Boston Scientific’s(NYSE:BSX) Watchman 1st-of-its-kind left atrial appendage (LAA) closure device for reducing the risk of stroke and systemic embolism.
The panel voted 13-1 in favor of safety, effectiveness and Watchman’s benefits outweighing its risks, with some modifications to Boston Scientific’s proposed indications for use, label and post-market study of the device.
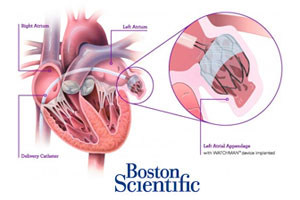
Watchman is a percutaneously delivered permanent cardiac implant, designed for implant in the heart’s left atrial appendage to prevent thromboembolism. Boston Scientific is seeking indication to treat patients with non-valvular atrial fibrillation who are eligible for warfarin therapy based on CHADS2 or CHA2DS2–VASc scores.
This is not Boston Scientific’s 1st swing at FDA approval for Watchman, and the FDA has in the past gone against the panel to turn down the Watchman application.
The FDA in March 2010 issued a Not Approvable letter for the device, ruling that existing data failed to demonstrate a reasonable assurance of safety and effectiveness. The agency ceded that the LAA closure device showed potential clinical benefit in the PROTECT AF trial, according to background materials released ahead of the meeting. The FDA’s previous decision against approval came despite a 7-5 vote for in favor of the device, with conditions, in April 2009 from the agency’s Circulatory System Devices Panel.
In refusing to approve Watchman’s initial application, the FDA cited a handful of problems with the PROTECT AF trial, including that the study enrolled a substantial number of study subjects who may have been acceptable candidates for aspirin therapy rather than anticoagulation; there was concomitant use of chronic clopidogrel therapy in both study groups (51% of follow-up time in device subjects and 16% of follow-up time in control subjects); and device implantation raised safety concerns such as pericardial effusion and air embolism.
Since then Boston Scientific has collaborated with the FDA to design the newer PREVAIL study clinical study, which aimed to address the limitations of the PROTECT AF data.
The FDA recognized the value in the safety and effectiveness information captured in PROTECT AF, and, in efforts to reduce the burden on the company, co-developed with Boston Scientific a Bayesian study for PREVAIL in which a portion of the PROTECT AF data could be used as an informative prior to collecting additional safety and effectiveness data on Watchman.
In addition to new data collected in PREVAIL, continued follow-up of PROTECT AF subjects was collected to provide critical insights into long-term device safety and effectiveness.
Today’s panel was tasked with discussing and voting on whether Watchman can now be approved based on the results of PREVAIL and additional long-term follow-up data from PROTECT AF, especially regarding the proposed indications for use and the proposed post-approval study. Data from the PREVAIL study hit 2 of the 3 primary endpoints for safety and effectiveness, but failed to demonstrate any improvement in rates of stroke, systemic embolism and cardiovascular or unexplained death at 18 months between patients who received the Watchman device and those who were treated only with warfarin, a blood thinner.

