ReCor Medical
Renal denervation isn’t dead, despite Medtronic trial’s failure
Hypertension: ReCor charges ahead with ultrasound renal denervation
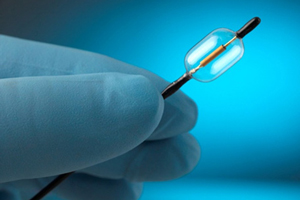
Hypertension: ReCor launches post-market study of next-gen Paradise system

California medical device startup ReCor Medical launched a post-market study of its next-generation Paradise renal denervation system in treatment of patients with resistant high blood pressure.
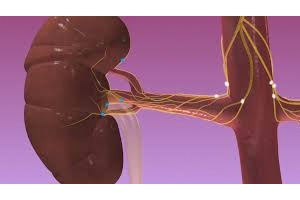
The 50-patient study has already enrolled patients at a participating facility in the Netherlands with the Paradise device, the only renal denervation device on the European market that treats high blood pressure with ultrasound rather than radiofrequency.
Hypertension: ReCor Medical lands Euro win for ultrasound-based renal denervation

European regulators granted CE Mark approval to California-based medical device startup ReCor Medical for its 2nd-generation Paradise system, which treats drug-resistant hypertension by targeting the renal nerves.
ReCor Medical adds 4 to expand its medical advisory board | Personnel Moves
FDA looks to strengthen device surveillance post-Riata | MassDevice.com On Call

MASSDEVICE ON CALL — Under increasing pressure from doctors, the FDA this month will unveil a new medical device post-market surveillance program aimed at better tracking potentially faulty devices.
The more rigorous program will include unique device identifiers that allow the FDA, doctors and manufacturers to track individual devices and better monitor, track and report defects.