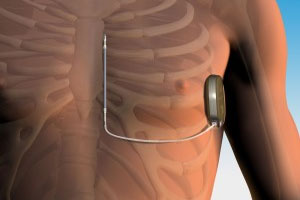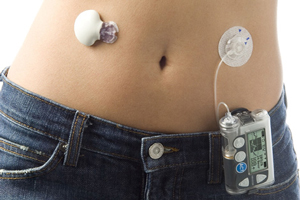
Medical device industry titan Medtronic (NYSE:MDT) filed a bid for FDA approval of its MiniMedR 530G system, which the company bills as the world’s first insulin pump that delivers and halts insulin automatically, based on sensor readings of a patient’s glucose levels.