
Medical devices maker Stryker (NYSE:SYK) isn’t downtrodden in light of the FDA’s decision to restrict use of the Wingspan brain stent system to a narrower group of patients – Stryker seems genuinely delighted.
The Medical Device Business Journal — Medical Device News & Articles | MassDevice

Westborough, Mass.-based Reflectance Medical became the first company to receive FDA 510(k) clearance for an oximeter and tissue perfusion system designed for patients with pigmented skin.
The company’s CareGuide oximeter continuously monitors muscle oxygen saturation (SmO2) and simultaneously assesses tissue perfusion in patients with darker skin tones.
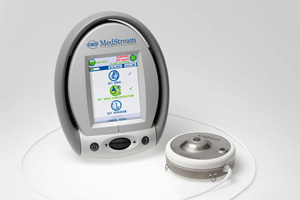
The FDA granted PMA supplemental approval to Codman & Shurtleff for the Medstream programmable infusion device, an implantable infusion pump and catheter used in treatment of movement disorders.
The Medstream system delivers continuous doses of an anti-spasm drug directly to he spinal canal to relieve spasticity, a disorder affecting more than 12 million people worldwide, which is often caused by stroke, cerebral palsy, multiple sclerosis or spinal cord injury, according to the company.

By Stewart Eisenhart, Emergo Group
The US Food and Drug Administration has opened small business qualification to foreign companies–including those that have not submitted US income tax returns–and also eliminated establishment registration fee discounts for small businesses as part of the agency’s updated Medical Device User Fees for 2013.

Top FDA officials were aware of the surveillance and monitoring programs targeting agency employees who raised questions about certain medial device review practices, according to a letter obtained by the Wall Street Journal.
Agency leadership, including Commissioner Dr. Margaret Hamburg and medical devices chief Dr. Jeffrey Shuren, were apprised of surveillance efforts against a group of medical device reviewers accused of leaking proprietary information to the media, according to the letter.
Sunshine Heart (ASX:SHC) won conditional approval from the FDA for the next generation of its flagship C-Pulse heart assist system, an outside-the-bloodstream left ventricular assist device that helps circulate blood and reduce the pumping burden on a patient’s heart.

Shares of NeuroMetrix (NSDQ:NURO) are soaring this morning on news of a U.S. regulatory win for its Sensus pain management system.
The FDA granted 510(k) clearance to the system but has yet to formally announce the decision, according to the watchdog agency’s website.
NURO shares surged as much as 22% this morning on the news and were up 10.8% to 72¢ as of about 10:20 a.m.
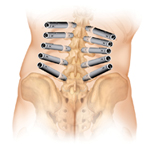
Johnson & Johnson’s (NYSE:JNJ) newly formed DePuy Synthes Spine division came out of the gates swinging, announcing its 1st regulatory win less than a month after closing the books on the $19.7 billion merger.

A member of the group of scientists suing the FDA for surveillance and alleged persecution was under increased scrutiny this week as breast imaging experts accused him of having blocked new breast cancer detection products from reaching the U.S. market.
Dr. Robert Smith spent nearly 4 years at the FDA’s radiological devices arm, during which time none of the 6 digital mammography systems up for approval passed muster with the federal watchdog agency.

