
Minnesota weight-loss devices maker EnteroMedics (NSDQ:ETRM) announced today that CFO and senior vice president Greg Lea had also been appointed chief operating officer.
The Medical Device Business Journal — Medical Device News & Articles | MassDevice

Minnesota weight-loss devices maker EnteroMedics (NSDQ:ETRM) announced today that CFO and senior vice president Greg Lea had also been appointed chief operating officer.

Shares of Stryker (NYSE:SYK) hit a 52-week high yesterday, peaking at $64.46 apeice before closing at $63.88, down 0.2% on the day.
STK shares are up about 3% since the medical device company reported 4th-quarter and 2012 results Jan. 24. Stryker said yesteday that it plans to issue a 26.5¢ dividend to shareholders.

Patients with Parkinson’s disease may benefit from receiving neurostimulating brain implants earlier in the course of the disease, according to researchers.
Treatment with neurostimulators, which delivery mild electrical pulses to targeted areas in the brain, are already standard for patients with advanced Parkinson’s disease, but researchers suggested that it may benefit patients "at an earlier stage than current recommendations suggest."

MASSDEVICE ON CALL — Massachusetts medical device titan Boston Scientific (NYSE:BSX) will need to add 50 new employees for each 500 square-feet of new space at its new Marlborough headquarters, in compliance with the 2nd phase of a long-term tax incentive program agreed upon by the city.
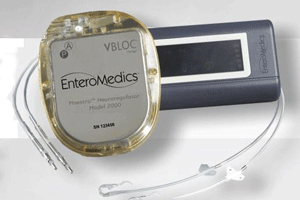
Wall Street fled in droves after weight-loss devices maker EnteroMedics (NSDQ:ETRM) announced that its Maestro vagal blocking system failed to meet endpoints in a pivotal study, but the mass exodus isn’t discouraging the company from moving forward with a bid for FDA approval.

Shares of Cyberonics (NSDQ:CYBX) are up some 6% today after the medical device company said an ex-sales rep voluntarily dismissed a wrongful termination lawsuit.
Andrew Hagerty sued the Houston, Texas-based company last August, alleging that Cyberonics canned him after he refused to sell unneeded replacement batteries for implanted pulse generators designed to treat epilepsy.

Minneapolis medical device company Medtronic’s (NYSE:MDT) neurostimulation implants, used in treatment of chronic pain, won European regulatory approval for compatibility with full-body MRI scanning.

Minneapolis medical device giant Medtronic (NYSE:MDT) launched a new global study of its Specify 5-6-5 spinal cord stimulation therapy in treatment of patients with failed back surgery syndrome and predominant low back pain.
The PROMISE study, which will enroll up to 300 patients, will evaluate treatment with Specify SCS and drug therapy and compare it to treatment with drug therapy alone.

SPR Therapeutics touted a regulatory win across the pond for its Smartpatch peripheral nerve stimulation system for treating chronic shoulder pain as a result of stroke.
Cleveland, Ohio.-based SPR’s Smartpatch system is a "skin-mounted external stimulator" that can deliver up to 30-days of pain relief, according to a press release.

Cleveland-area startup SPR Therapeutics closed a $7.8 million Series A financing round to commercialize its neurostimulation pain relief device.
The funding came from NDI Healthcare Fund, Public Square Partners and individual investors, according to a press release.

