
Replacement heart valves: Real-world results for Sapien equivalent to trial results

The Medical Device Business Journal — Medical Device News & Articles | MassDevice


MASSDEVICE ON CALL — Medical device titan Medtronic (NYSE:MDT) announced late last week that the FDA had put its highest-risk label on a company recall of certain guidewires. The agency deemed the recall a "Class I" issue, reserved for device defects that could "cause serious adverse health consequences or death."

Lumenis Ltd. announced today regulatory wins in the U.S. and Japan for its Array LaserLink pattern scanning device for multi-spot laser photocoagulation for use during retinal surgery.
The Array LaserLink, which turns an "examination slit lamp" into a medical laser, expands on the Lumenis laser system to deliver “pattern scanning capabilities, aimed to provide greater predictability in retinal treatments and presumably less collateral damage to the retina,” according to a press release.

The FDA this week issued its final rule on scleral plugs, down-classifying them to a lower-risk category of medical devices in need of less stringent review.
FDA regulators today announced that they granted premarket approval for NeuroPace for its anti-epilepsy device, an implantable neurostimulator that helps prevent seizures.
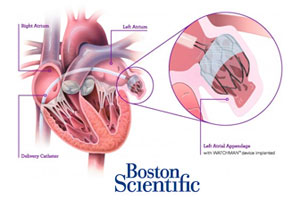
Boston Scientific (NYSE:BSX) won a date with an FDA advisory panel for its Watchman heart implant after the anti-stroke device met 2 of 3 endpoints in a clinical trial.

Cochlear Ltd. (ASX:COH) said an advisory committee to the FDA this week voted favorably on the Nucleus Hybrid L24 implant system, which combines acoustic amplification of low frequencies and electrical stimulation for high frequencies in a single device.

The FDA put a Class I label on Baylis Medical’s recall of its TorFlex transseptal guiding sheath kit, the federal watchdog agency announced.
The recall was issued because the sheath could remove particulate of less than 5mm from the dilator and the particulate could end up in the bloodstream, the FDA said.

FDA regulators plan to meet next month to review a handful of medical devices and possibly recommend reclassification for spinal fusion ‘spheres’ and both stair-climbing and mechanical wheelchairs.
The agency noted in the Federal Register that its Orthopaedic & Rehabilitation Devices Panel will meet December 12 to review the technologies based on new reviews of their safety and effectiveness.


Topera said it landed a pair of regulatory wins for its FIRMap catheter.
The Palo Alto, Calif.-based medical device company said it won the CE Mark approval in the European Union and 510(k) clearance from the FDA for the device.

