Boston Scientific (NYSE:BSX) won a date with an FDA advisory panel for its Watchman heart implant after the anti-stroke device met 2 of 3 endpoints in a clinical trial.
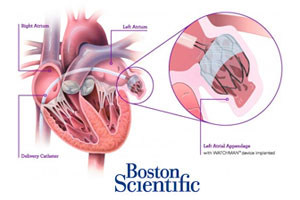
The Watchman implant, already available in 30 countries, is introduced via catheter and threaded into the heart to close off the left atrial appendage and capture any blood clots that may form there.
Today the FDA’s Circulatory System Devices Panel said it plans to convene Dec. 11 to discuss whether the Watchman device is safe and effective enough to recommend that the watchdog agency approve the device. Although the FDA is not bound by the recommendations of its advisory panels, it often follows their lead.
Will the FDA approve the Watchman implant? Tell us what you think
In March, Natick, Mass.-based Boston Scientific unveiled results from the Prevail clinical trial of the Watchman device, saying it met 2 of 3 co-primary endpoints for safety and effectiveness compared with drug therapy.
Watchman achieved its 2 safety endpoints, but researchers found no differences in rates of stroke, systemic embolism and cardiovascular or unexplained death at 18 months between patients who received the Watchman device and those who were treated only with warfarin, a blood thinner.
The device had more success in demonstrating lowered rates of acute death or serious vascular complications related to the procedure in the 7 days following implantation, as well as low rates of ischemic stroke or systemic embolism more than 7 days and up to 18 months following implantation, the company said at the time. Other research showed that Watchman may eliminate the need for long-term use of blood-thinning medications like warfarin.
The Prevail trial was launched in order to address safety concerns raised by a previous Watchman trial, Boston Scientific noted. The Protect AF trial showed that treatment with the Watchman device reduced a patient’s risk of hemorrhagic stroke compared with warfarin, but also showed an increase in procedure-related adverse events.
Boston Scientific acquired the Watchman device along with AtriTech for up to $375 million in 2011. Last summer the company closed enrollment in a 2nd trial of the device, with more than 2,000 subjects signing on for the randomized comparison of Watchman with warfarin in high-risk patients with atrial fibrillation. In August the device won expanded CE Mark approval in the European Union for the catheter-based clot-capture system, to patients with atrial fibrillation who can’t be treated with warfarin or new oral anticoagulants.

