Medtronic (NYSE:MDT) subsidiary CoraValve Inc. is asking a federal circuit court to throw out a $73.5 million jury verdict won by Edwards Lifesciences Inc. (NYSE:EW), arguing that its device does not infringe on a patent held by the Irvine, Calif.-based company due to on exact wording in the patent.
Replacement Heart Valves
Stents: Drug-eluters beat bare metal for patients over 85

Drug-eluting stents dealt another blow to their bare-metal cousins by leading to fewer adverse events post-procedure in patients over the age of 85 undergoing percutaneous coronary intervention, new research shows.
Patient with drug-eluting stents showed a 29% mortailty risk, compared with a 38% risk in patients with bare-metal stents, with the difference narrowing with increasing age, according to the study, published in the Journal of the American College of Cardiology.
Mayo study: TAVI learning curve is steep
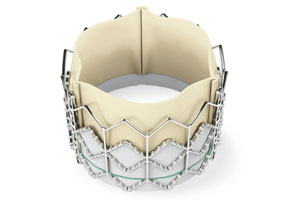
Edwards Lifesciences’ Sapien valve.
A new Mayo Clinic study suggests that it may take as many as 30 procedures for a physician to become proficient at transcatheter aortic valve implantation, rekindling the debate over what credentials docs ought to have to perform the procedure.
MassDevice Top 10 of 2011 | The top medical device stories of the year, Part I

Welcome to MassDevice’s annual audit of the ups, downs and in-betweens of the year that was. For the medical device industry, 2011 was more or less defined by a nagging sense of uncertainty that hung over the world’s med-tech companies like a grey cloud above a summer picnic.
But what did we learn from all the conjecture? In truth, not much. The year ends much as it began, full of questions with no cut and dry answers as we turn the calendar over into the new year.
Philips lands 510(k) for HeartNavigator valve replacement | Regulatory Roundup
Updated Dec. 23, 2011, to correct that the HeartNavigator is inserted through a small incision in the leg, not in the chest.
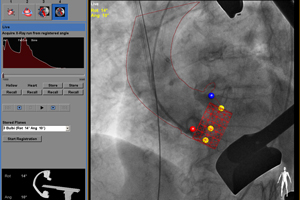 Philips’ HeartNavigator aortic valve replacement system uses x-ray imaging to guide the valve to the heart.
Philips’ HeartNavigator aortic valve replacement system uses x-ray imaging to guide the valve to the heart.Philips Healthcare (NYSE:PHG) landed FDA clearance for its HeartNavigator heart valve replacement system.
Medical City Dallas Hospital became the first U.S. hospital to use the system, which was launched in Europe in the first quarter of this year.
Three questions for Edwards Lifesciences CEO Michael Mussallem

If the good people of Orange County felt a gale force wind a few weeks back, it may have been a sigh of relief coming from the Irvine, Calif. headquarters of Edwards Lifesciences Corp. (NYSE:EW) after the FDA finally gave a much-anticipated green light to the company’s Sapien transcatheter aortic valve.
Edwards had been hanging fire since July, when an FDA advisory panel recommended that the federal watchdog agency approve the device.
TCT 2011: Medtronic’s Resolute DES just as effective as Abbott’s market-leading Xience V
TCT 2011: Benefits for Edwards’ valve patients increase at the two-year mark
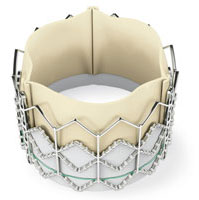
Patients treated with Edwards Lifesciences’ (NYSE:EW) Sapien heart valve had a lower mortality rate and spent less time in hospitals at the two-year mark, according to the data released at this year’s Transcatheter Cardiovascular Therapeutics symposium.
The Partner trial consisted of patients with aortic stenosis, randomized to receive a Sapien valve with transcatheter aortic valve replacement surgery or traditional open-heart surgery.
MassDevice.com +3 | Baxter’s CEO reaps $14M, Sapien trial cut short on adverse events, Devices plus drugs pose great potential

Say hello to MassDevice +3, a bite-sized view of the top three med-tech stories of the day. This latest feature of MassDevice.com’s coverage highlights our three biggest and most influential stories from the day’s news to make sure you’re up to date on the headlines that continue to shape the medical device industry.
If you read nothing else today, make sure you’re still in the know with MassDevice +3.
Update: Sapien transapical trial spiked on adverse events
Updated November 9, 2011 at 5:30 p.m. to reflect that the trial was conducted by a Danish team, not by Edwards Lifesciences.

Sapien
Researchers cut short a trial of Edwards Lifesciences (NYSE:EW) Sapien aortic valve replacement device due to adverse events associated with the implant, according to a securities analyst.



