
Abiomed (NSDQ:ABMD) said the Centers for Medicare & Medicaid Services approved a trio of reimbursement codes for its Impella heart pumps, allowing ABMD shares to regain some of the roughly 30% investors pared from the stock this week.
The Medical Device Business Journal — Medical Device News & Articles | MassDevice
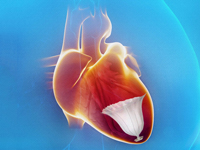
California-based medical device company CardioKinetix landed CE Mark approval in the European Union for the full suite of its Parachute ventricular partitioning devices, including 8 sizes of the device designed to provide support to weakened heart muscles. .

The Centers for Medicare & Medicaid Services are likely to leave current payment intact for left ventricular assist devices, but an agenda released ahead of a November meeting hints at possible restrictions on off-label use for the costly devices.
The federal agency plans to meet November 14 to review available evidence on LVADs and discuss how best to optimize outcomes for heart failure patients, especially when it comes to pinpointing which patients are likely to benefit from the procedure.

The European Union granted CE Mark approval to a pair of medical device companies, CircuLite and NeuroSigma, for their technologies designed to treat heart failure, epilepsy and depression.
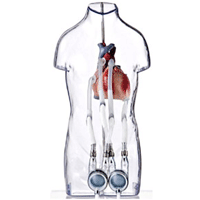
Children with heart failure survived longer while awaiting a heart transplant when they received therapy with Berlin Heart’s Excor pediatric ventricular assist device than when treated with the current standard of care, according to a new study.
The Excor PAD device was not without risks, but survival rates for children on the device were "significantly higher" than those in the control group, according to the study’s authors.

Even though 2nd-quarter net losses soared for HeartWare International (NSDQ:HTWR), sales rose 43% for the medical device company, sending shares up a tick today.
HeartWare, which makes implantable heart pumps, reported losses of $22.8 million, or $1.61 per share, on sales of $29.1 million during the 3 months ended June 30.


Say hello to MassDevice +3, a bite-sized view of the top three med-tech stories of the day. This feature of MassDevice.com’s coverage highlights our 3 biggest and most influential stories from the day’s news to make sure you’re up to date on the headlines that continue to shape the medical device industry.
If you read nothing else today, make sure you’re still in the know with MassDevice +3.

