
Billionaire hedge fund manager David Tepper, of Appaloosa Management, bought 7.8 million shares of Boston Scientific stock during the 4th quarter, according to regulatory filings, for a stake with a market value of $41.6 million as of Dec. 31.
The Medical Device Business Journal — Medical Device News & Articles | MassDevice

Billionaire hedge fund manager David Tepper, of Appaloosa Management, bought 7.8 million shares of Boston Scientific stock during the 4th quarter, according to regulatory filings, for a stake with a market value of $41.6 million as of Dec. 31.
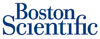
Boston Scientific’s (NYSE:BSX) Ion and Taxus Liberté drug-eluting stents won pre-market approval from the FDA to treat heart attack patients.
The Natick, Mass.-based med-tech maker said the devices, which elute the anti-restenosis drug paclitaxel, are the only DES approved to treat patients with acute myocardial infarction.
The stents are fashioned from Boston Scientific’s next-generation platinum-chromium alloy. The company said 10% of all coronary interventions are performed for patients having a heart attack.

Although his 10-year tenure at the helm of Johnson & Johnson (NYSE:JNJ) is likely to be remembered for the spate of recalls that have bedeviled the healthcare giant for the past few years, William Weldon should also be remembered for something he didn’t do: Spend billions on a buyout of pacemaker company Guidant Corp.
Medtronic landed a significant win with the announcement of an FDA win for its Resolute Integrity drug-eluting stent, the first device on the U.S. market cleared for patients with coronary artery disease as well as diabetes.
Diabetic patients make up about ⅓ of all those with CAD, and they represent a difficult-to-treat population.

Say hello to MassDevice +3, a bite-sized view of the top three med-tech stories of the day. This feature of MassDevice.com’s coverage highlights our 3 biggest and most influential stories from the day’s news to make sure you’re up to date on the headlines that continue to shape the medical device industry.
If you read nothing else today, make sure you’re still in the know with MassDevice +3.
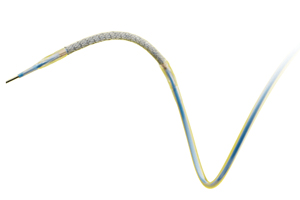
The FDA’s neurological devices panel will review clinical data from trials of Stryker’s (NYSE:SYK) Wingspan brain stent system next month, aiming to address harsh criticisms and public petitions urging the agency to recall the device.
The panel plans to consider pre-market and post-market data about the device, including the Gateway PTA balloon catheter and stent delivery system, to address concerns that Wingspan may lead to heightened risk of stroke or death.

Say hello to MassDevice +7, a bite-sized view of the top seven med-tech stories of the week. This latest feature of MassDevice.com’s coverage highlights our seven biggest and most influential stories from the week’s news to make sure you’re up to date on the headlines that continue to shape the medical device industry.
If you read nothing else this weekend, make sure you’re still in the know with MassDevice +7.
Boston Scientific (NYSE:BSX) landed CE Mark approval in the European Union for its Infinion 16 percutaneous lead, designed for use with the company’s Precision Plus spinal cord stimulator system.
BSX’s spinal cord stimulator leads are designed to mask pain signals to the brain by delivering electrical pulses from an implantable pulse generator.

MASSDEVICE ON CALL —A clutch of 26 states arguing against the individual insurance mandate in President Barack Obama’s health care reform argued before the Supreme Court that the measures represented an abuse of Congressional power.

