
Boston Scientific (NYSE:BSX), stung by recent studies showing that the thinner design of next-generation stents can deform during deployment, is on the offensive with an new video.
The Medical Device Business Journal — Medical Device News & Articles | MassDevice

Boston Scientific (NYSE:BSX), stung by recent studies showing that the thinner design of next-generation stents can deform during deployment, is on the offensive with an new video.

Wall Street’s overall take on sales and earnings forecasts for medical device makers this year is too high, according to Goldman Sachs (NYSE:GS) analyst David Roman, who sees The Street’s 4th-quarter top-line estimates as split evenly among companies with upside and downside.

Next-generation drug-eluting stents proved safer than their predecessors and their bare-metal cousins for rates of restenosis, stent thrombosis and subsequent death, flipping the tables on previous findings.
New results from Swedish Coronary Angiography & Angioplasty Registry data taken between Nov. 2006 and Oct. 2010 show that DES have finally taken the lead for safety in the stent arena.
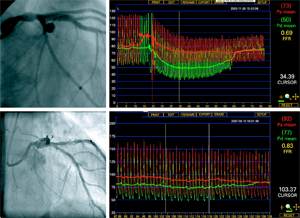
St. Jude Medical (NYSE:STJ) said it’s halting its Fame II trial after an independent safety advisory board recommended the trial be stopped due to strong positive results.

Say hello to MassDevice +3, a bite-sized view of the top three med-tech stories of the day. This feature of MassDevice.com’s coverage highlights our 3 biggest and most influential stories from the day’s news to make sure you’re up to date on the headlines that continue to shape the medical device industry.
If you read nothing else today, make sure you’re still in the know with MassDevice +3.
Updated 4:45 p.m. with competing device information at the bottom
Boston Scientific’s (NYSE:BSX) Epic stent system knocked its primary 9-month endpoints out of the park, according to data from the company’s Orion trial.
The company beat its target 17% goal for major adverse events associated with iliac stenting, touting a 3.4% MAE rate over 9 months for the "intent to treat" population in the study.

Say hello to MassDevice +3, a bite-sized view of the top three med-tech stories of the day. This latest feature of MassDevice.com’s coverage highlights our three biggest and most influential stories from the day’s news to make sure you’re up to date on the headlines that continue to shape the medical device industry.
If you read nothing else today, make sure you’re still in the know with MassDevice +3.
A deep brain stimulation implant made by St. Jude Medical’s (NYSE:STJ) proved effective in the treatment of patients Parkinson’s disease, according to results published in The Lancet.
The 136-patient study compared patients suffering from Parkinson’s who were implanted with St. Jude’s Libra and LibraXP DBS systems with and without stimulation. The study enrolled patients who had suffered from Parkinson’s for at least 5 years, or who had 6 hours a day with diminished motor symptom control and muscle ticks.
Boston Scientific Corp. (NYSE:BSX) is working to pare between $650 million and $750 million worth of costs from its budget in a quest for double-digit earnings growth, according to interim CEO Hank Kucheman.
The cuts take into account an estimated $150 million hit from the medical device tax,which is slated to go into effect next year, Hank Kucheman said at the J.P. Morgan health care conference today.

Eric Bouvier took over the corner office at BG Medicine (NSDQ:BGMD) yesterday as president & CEO of the Waltham, Mass.-based diagnostics developer.
Prior to joining BG, Bouvier, 51, headed the Immunoassay business at global diagnostics company BioMérieux. He also held the role of president & CEO of BioMérieux’s U.S. subsidiary bioMerieux Inc.

