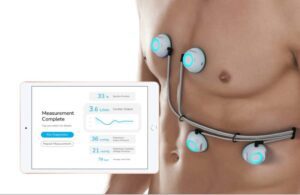
Los Angeles-based Sensydia designed the AI-based, non-invasive CPS to use heart sound analysis. It may enable earlier detection and improved therapy guidance for heart failure and pulmonary hypertension patients.
CPS uses ultra-sensitive biosensors for the rapid, non-invasive measurement of a number of cardiac metrics. These include ejection fraction, cardiac output, pulmonary artery pressure and pulmonary capillary wedge pressure. It all comes together in one handheld device, Sensydia said.
Normally, patients must undergo echocardiography and invasive catheterization to obtain these measurements. CPS offers fast and safe measurements and can be performed almost anywhere with minimal training, according to a news release.
Sensydia conducted the study at the University of Pittsburgh Medical Center (UPMC).
“When we began this study at UPMC during the height of the COVID-19 pandemic, our original enrollment target for the study was 110 subjects, but we ended up doubling that after hearing positive feedback from the UPMC study staff,” said Anthony Arnold, president and CEO of Sensydia. “This is Sensydia’s fourth successful study and we will continue to collect data across multiple sites to improve the performance and utility of the artificial intelligence algorithms that power our breakthrough CPS platform.”
Sensydia previously conducted a study at the Ronald Reagan UCLA Medical Center. That study contributed to FDA clearance for its ejection fraction algorithm. It conducted another study at the OHSU Knight Cardiovascular Institute for its cardiac output algorithm.
The FDA granted CPS breakthrough device designation in January 2022. Sensydia plans to use data from the 225-patient study to develop its CPS pulmonary pressure algorithms. It also said it is closing a new round of funding at this time.

