Covidien (NYSE:COV) announced a new recall, its 2nd in as many months, after receiving reports of serious burn injuries associated with certain grounding pads contained in ablation device kits.
The medical device maker recalled the pads and the Cool-tip RFA ablation systems that contain them after at least 4 patients experienced "serious injuries, burns at the pad site," according to the company.
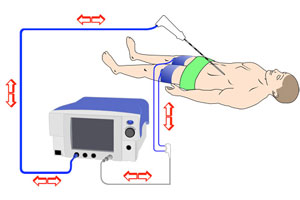
The Cool-tip RFA system is an ablation device that may be used as an alternative to surgery for some cancers in the liver or other soft tissue, according to Covidien’s website. The device in May 2006 won FDA clearance for use in non-resectable liver tumors.
The Cool-tip RFA delivers radiofrequency energy to heat and destroy target tissue, either through open surgery, percutaneously or laparoscopically based on a physician’s preference. The 4 single-use grounding pads go across the thighs.
Covidien recalled certain lots of the pads and the Cool-tip RFA kits manufactured prior to August 30, 2011, according to the recall notice.
That’s bad news for the company, which earlier this month recalled certain lots of its cuffed Shiley tracheostomy tubes after receiving reports that the devices malfunctioned during patient ventilation.
And last month the Mansfield, Mass.-based medical device maker issued a renewed U.K. warning on potential battery defects in certain mobile ventilators used primarily by patients in their homes.
Earlier in July the FDA issued a safety alert after receiving reports of more than 100 adverse events, including 9 patient deaths, that may have been caused by Onyx brain blood vessel plugs made by Covidien subsidiary ev3 Inc.
In June the FDA asked Covidien to defend its management of the recall of its Duet TRS surgical staples, which were pulled off the shelves after they were implicated in 13 patients injuries and 3 deaths.
COV shares slid slightly today, lower by 0.7% and trading at $56.85 as of about 2:45 p.m.

