
By Stewart Eisenhart, Emergo Group
Brazilian regulators have issued new requirements under Normative Instruction 9/2013 that expand certification requirements for some electronic and other medical devices.
The Medical Device Business Journal — Medical Device News & Articles | MassDevice
FDA | Recalls | 510(k) | Pre-Market Approvals | MDSAP | Clinical Trials

By Stewart Eisenhart, Emergo Group
Brazilian regulators have issued new requirements under Normative Instruction 9/2013 that expand certification requirements for some electronic and other medical devices.
Medtech titan St. Jude Medical (NYSE:STJ) won FDA premarket approval for its Cool Flex cardiac ablation system with indication to treat irregular heart rhythms.

The U.S. Senate yesterday approved an omnibus, $1.012 trillion budget bill that includes the restoration of some $85 million in user fees from medical device and pharmaceutical companies that was held in abeyance by sequestration.
President Barack Obama, who has until midnight tomorrow to sign the measure into law, is expected to quickly add his signature after the 72-26 vote in the Upper Chamber.

By: Margaret A. Hamburg, M.D.
Ensuring the safety of the food supply and the safety and effectiveness of drugs, devices and biologics has always been at the core of FDA’s responsibility to protect the public health – and always will be.


MASSDEVICE ON CALL — Medtech industry lobbying group AdvaMed offered high praise for the federal government’s 2014 omnibus appropriations legislation, which restored important funding for the FDA.
The bill returned to the agency about $85 million in collected user fees that had been locked away due to the federal sequester, funds that medical device and drug companies pay for applications for FDA review.
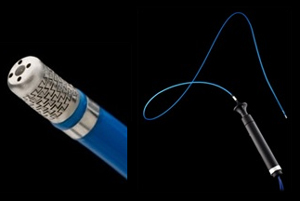
MediValve said it won both CE Mark clearance in Europe and FDA 510(k) clearance in the U.S., for its acWire Guidewire.
Kibbutz Hama’apil, Israel-based MediValve started in 2010, as a portfolio company of The Trendlines Group. The company’s only product so far is the acWire.
The acWire Guidewire is a single-use device that acts as a positioning tool for peripheral vascular and heart catheterization procedures, for diagnostic and interventional devices. Landing both clearance in the U.S. and Europe is a big win for the small company, which means it can be marketed in both areas. No indication has been made on this front, according to a press release.

The FDA this month finalized a rule requiring more pediatric patient information from medical devices going through the premarket approval pathway, even if the products are intended for adults.


