In a speech on the Senate floor this week, Sen. Chuck Grassley (R-Iowa) blasts the FDA over reports that the agency surveiled scientists in its medical device review section.
Food & Drug Administration (FDA)
Second Sight lands FDA panel date for retinal prosthesis HDE

Second Sight Medical Products, the world’s only developer and manufacturer of an approved retinal prosthesis for the blind, won a date with the FDA to consider humanitarian device exemption status for its Argus II system.
On Sept. 28, 2012, the FDA’s ophthalmic devices panel will review the application for the Argus II device for patients aged 25 and older who have severe to profound retinitis pigmentosa, experience bare or no light perception in both eyes and have "previous history of useful form vision."
Reports: FDA surveilled dissident scientists

An FDA probe of 5 dissident scientists from its medical device review arm may have broken the law when it resulted in the inadvertent public disclosure of confidential information.
MassDevice.com +7 | The top 7 med-tech stories for the week of July 9, 2012

Say hello to MassDevice +7, a bite-sized view of the top seven med-tech stories of the week. This latest feature of MassDevice.com’s coverage highlights our seven biggest and most influential stories from the week’s news to make sure you’re up to date on the headlines that continue to shape the medical device industry.
If you read nothing else this weekend, make sure you’re still in the know with MassDevice +7.
FDA’s user fees not a sure win for medical devices | MassDevice.com On Call

MASSDEVICE ON CALL — Medical device makers may still have a way to go in ensuring the increased speed and efficiency they were promised when they signed off on the latest iteration of the FDA’s user fee program.
The program, which began in 2003 has returned "mixed results," according to Forbes writer John Graham, who conducted an analysis for the Pacific Research Institute of the FDA’s expanded budget and subsequent changes in productivity.
Asthmapolis wins FDA clearance for new sensor and software | Regulatory Roundup
FDA releases 180-day PMA memos

The FDA’s Center for Devices & Radiological Health released a selection of summary review memos detailing applications to make substantial changes to already-approved medical devices.
The federal watchdog agency’s med-tech oversight arm said it began releasing the documents as part of the CDRH’s transparency initiative.
The 180-day supplements seek to make "a significant change in components, materials, design, specification, software, color additive, and labeling to an approved premarket application or premarket report," according to the agency.
MassDevice.com +3 | The top 3 med-tech stories for July 10, 2012

Say hello to MassDevice +3, a bite-sized view of the top three med-tech stories of the day. This feature of MassDevice.com’s coverage highlights our 3 biggest and most influential stories from the day’s news to make sure you’re up to date on the headlines that continue to shape the medical device industry.
If you read nothing else today, make sure you’re still in the know with MassDevice +3.
St. Jude wins 510(k) for ViewFlex Xtra ultrasound catheter
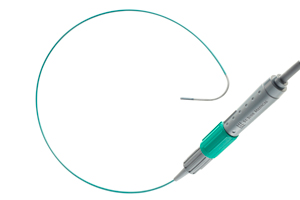
St. Jude Medical (NYSE:STJ) touted 510(k) clearance for its ViewFlex Xtra intracardiac echocardiography catheter for use with the device giant’s ViewMate Z ultrasound console.
China regulators warn on Micro-Pace pacemakers | MassDevice.com On Call

MASSDEVICE ON CALL — China’s Dept. of Health warned local healthcare providers of potential sensing issues noted in dual-chamber temporary pacemakers made by Pace Medical.
The warning involves Micro-Pace Model 4580 systems, programmable, multi-mode, multi-parameter cardiac pacemakers made by San Diego-based Pace Medica. The Micro-Pace devices also have FDA approval in the U.S.
MassDevice.com +7 | The top 7 med-tech stories for the week of July 2, 2012

Say hello to MassDevice +7, a bite-sized view of the top seven med-tech stories of the week. This latest feature of MassDevice.com’s coverage highlights our seven biggest and most influential stories from the week’s news to make sure you’re up to date on the headlines that continue to shape the medical device industry.
If you read nothing else this weekend, make sure you’re still in the know with MassDevice +7.



