
The FDA’s Radiological Devices Panel this week voted to grant Hologic’s (NSDQ:HOLX) expanded approval for its 1st-of-its-kind Selenia Dimensions 3D digital mammography system.
The Medical Device Business Journal — Medical Device News & Articles | MassDevice

NuVasive Inc. (NSDQ:NUVA) said the FDA granted pre-market approval for its PCM cervical disc implant, sending NUVA shares up more than 2% on Wall Street today.
The San Diego-based medical device company said the implant is designed to preserve motion in the cervical spine, in lieu of a standard fusion procedure. The FDA nod came after a 403-patient investigation device exemption trial, according to NuVasive.
Israeli device maker InSightec won FDA approval for its ExAblate MRI-guided focused ultrasound therapy with indication to treat pain resulting from bone metastases in patients who failed or aren’t good candidates for radiation therapy.
Bone metastases occur when cancer cells break off from the primary tumor site and spread to other parts of the body, commonly leaving patients with significant physical and emotional pain, according to a press release.

Edwards Lifesciences (NYSE:EW) won FDA approval to market its Sapien transcatheter aortic valve implantation system to lower-risk patients, despite concerns about limited patient data on the long-term side effects of the device.

Edwards Lifesciences (NYSE:EW) won FDA approval to market its Sapien transcatheter aortic valve implantation to lower-risk patients, despite concerns about limited patient data on the long-term side effects of the device.
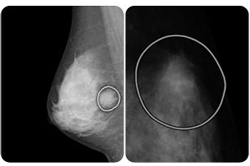
Medical imaging analysis devices maker VuCOMP touted FDA premarket approval for its new M-Vu system for helping radiologists detect breast cancer earlier.
The Version 2 M-Vu system’s new indication allows the company to market its devices as an computer-aided detection adjunct to digital mammography.
Paradigm Spine won FDA approval for its Coflex spinal stenosis treatment system, the 1st such device to treat moderate to severe cases of spinal stenosis that may present with or without back pain.
Paradigm calls the Coflex device the "the 1st and only motion-preserving alternative to fusion for the treatment of moderate to severe spinal stenosis."

HeartWare International (NSDQ:HTWR) decided to back out of the REVIVE-IT study of its left-ventricular assist device, citing the need to focus efforts on the company’s efforts to win FDA approval for the device.
The REVIVE-IT study, which the HeartWare had contributed about $600,000 to, was designed to evaluate the company’s LVAD system in patients with lower-risk heart disease.

Federal regulators issued new guidelines for medical device makers seeking pre-market approval under the FDA’s new user fee program.
The FDA’s Center for Devices & Radiological Health this week issued new guidance on how the agency hopes to hit the goals for medical device review that were agreed to in the latest generation of the Medical Device User Fee & Modernization Act.

Privately held CoAxia Inc. landed a date with the FDA’s Neurological Devices Panel to submit its NeuroFlo catheter for de novo approval.
The Minneapolis-based company’s NeuroFlo device, which already has CE Marking for the European Union, aims to divert blood flow in the brain for patients suffering cerebral ischemia as a result of stroke, vasospasm, or other conditions.
The device boasts 35-50% improvement in cerebral perfusion and can be placed in 10-15 minutes via the abdominal aorta, according to the company’s website.

The FDA’s Center for Devices & Radiological Health released updated summary review memos detailing applications that made substantial changes to already-approved medical devices.
The 180-day supplements seek to make "a significant change in components, materials, design, specification, software, color additive, and labeling to an approved premarket application or premarket report," according to the agency.

