Intact Vascular said yesterday that it named medical device veteran Bruce Shook as its new president & CEO.
VuComp Inc.
Press Release: VuCOMP Announces FDA Approval of M-Vu Computer-Aided Detection (CAD) for Mammography Version 3.1

VuCOMP lands FDA nod for updated M-Vu mammography system

Cancer detections systems maker VuCOMP announced this week that it won FDA approval for new computer-aided detection software, launching version 3.1 of its M-Vu CAD for mammography.
VuComp wins FDA clearance for breast density measurement tool

VuComp said it won 510(k) clearance from the FDA for its M-Vu breast density tool, which provides automated assessment of breast density from a digital mammogram.
The M-Vu uses advanced computer vision algorithms to evaluate the appearance of structures and textures in the breast to differentiate between fatty and dense regions. It then calculates a percentage of breast area that is dense, translating it to 1 of 4 density categories corresponding to the BI-RADS standard.
Breast cancer: VuCOMP wins expanded PMA for digital mammogram analysis
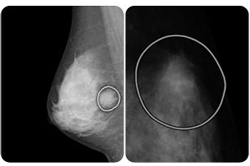
Medical imaging analysis devices maker VuCOMP touted FDA premarket approval for its new M-Vu system for helping radiologists detect breast cancer earlier.
The Version 2 M-Vu system’s new indication allows the company to market its devices as an computer-aided detection adjunct to digital mammography.
Medtronic lands FDA clearance, plans launch for next-gen CRM lead | Regulatory Roundup

Medtronic Inc. (NYSE:MDT) landed FDA clearance for its DF4 high-voltage right ventricular lead for use with implantable cardiac rhythm management devices.
The DF4, which builds on Medtronic’s Sprint Quattro lead, aims to help simplify the implantation process with a redesigned port and by providing visual confirmation of the lead’s connection to the device.


