Nevro Corp. said it plans to raise more than $100 million in an initial public offering of stock for its implantable spinal cord stimulation system to treat chronic pain.
Spine
Judge upholds $30M infringement ruling against NuVasive, cancels trademark
VertiFlex lands FDA panel date for Superion interspinous spacer
VertiFlex Inc. won a date with an FDA advisory panel to review the data behind its Superion interspinous spacer ahead of its bid for pre-market approval from the watchdog agency.
Baxano puts itself on the auction block
Intrinsic Therapeutics raises $17.2M for spinal surgery devices

Massachusetts-based Intrinsic Therapeutics is nearing the close of a $20.9 million equity round, with about $17.3 million raised so far.
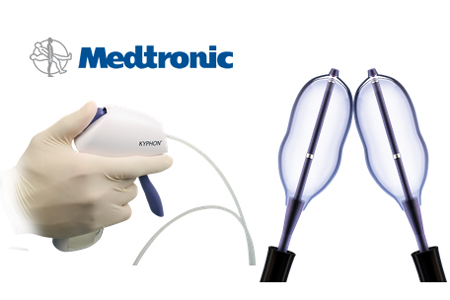
Medtronic unveils next-gen balloon kyphoplasty
Medtronic allies with Ortho Kinematics for spinal diagnostic test
ReWalk Robotics prices IPO at $36M, below range
ReWalk Robotics (NSDQ:RWLK) priced its initial public offering below its forecast range, at $12 per share, grossing some $36.0 million for its exo-skeleton suit.
DoJ says recorded meeting uncovers kickback scheme at Reliance Medical

Federal investigators are pushing forward with claims against Reliance Medical Systems, saying that they have evidence that the company’s owners devised a kickback scheme to induce doctors to use Reliance spinal implants, even sometimes when patients didn’t need the procedure.
Mazor Robotics wins China OK for Renaissance robotic surgery device
Titan Spine’s new warranties cover revision surgery for spinal implants

Titan Spine announced this week that it’s offering a new series of warranties for its Endoskeleton line of interbody spinal fusion implants, promising a 1-time free revision surgery within 5 years.