Oncology
MassDevice Podcast: Mela Sciences’ CEO on the MelaFind odyssey
Boston Scientific launches nerve stimulation trial | MassDevice.com On Call

MASSDEVICE ON CALL —Boston Scientific Corp. (NYSE:BSX) enrolled the first patients into the NECTAR-HF clinical study testing chronic vagal nerve stimulation as a therapy for heart failure patients.
Spare the knife, keep the rod
Who will tell the men?
That’s what I wondered last week when I read the abstract about prostate cancer patients’ grim prognosis for normal sexual activity after surgery or radiation for their disease. The study appeared in the Journal of the American Medical Association.
Delcath’s Chemosat gives terminal cancer patients several more months of disease-free life
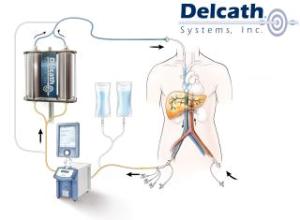
Delcath Systems Inc.’s (NSDQ:DCTH) Chemosat organ-targeting chemotherapy system extended the lives of terminal eye cancer patients by several months without disease progression, researchers said.
Melanoma of the eye often spreads to the liver, generally killing a patient in 1 to 2 months, according to a press release. Patients who received targeted treatment to saturate the liver lived an average of 8.1 months, compared to 1.6 months for patients receiving the best alternative care.
Delcath’s Chemosat fives terminal cancer patients several more months without disease progression
Contact: Emma Mason
wordmason@mac.com
ECCO-the European CanCer Organisation
Infusing chemotherapy into the liver gives extra months of disease-free life in melanoma patients
Stockholm, Sweden: Melanoma of the eye (ocular or uveal melanoma) frequently spreads to the liver and, once this has happened, there is no effective treatment and patients die within an average of two to four months. Only about one in ten patients live for a year. Now, final results from a phase III study have demonstrated that a new treatment significantly extends the time patients can live without the disease progressing.
Mela Sciences shares nearly double on news of FDA progress | MassDevice.com On Call

MASSDEVICE ON CALL — Mela Sciences (NSDQ:MELA) is on a roll this month, receiving an "approvable letter’ from the FDA just weeks after winning CE Mark approval in the European Union for its handheld skin cancer detector, MelaFind.
MELA shares shot up to $6.00 in early morning trading today, an 89 percent hike on yesterday’s close at $3.18. Shares had settled down to about $5.36 by mid-morning, still a 68 percent spike from yesterday.
Neoprobe executives plan stock sell-off if FDA green-lights Lymphoseek

Neoprobe Corp. (NYSE Amex:NEOP) are prepping to sell off about 500,000 shares of stock should the company’s new drug application get the green light from the FDA.
Senior vice president & CFO Brent Larson, regulatory affairs & quality assurance VP Rodger Brown and former president & CEO David Bupp revealed plans to sell substantial portions of their stakes at various times over up to one year.
Eliminate the 2.3 percent med-tech excise tax, urges Congresswoman Cathy McMorris Rodgers (R-Wash.) at Jobs & Innovation Forum | MassDevice.com On Call

MASSDEVICE ON CALL — Congresswoman Cathy McMorris Rodgers (R-Wash.) urged fellow Republicans to push for the repeal of the 2.3 percent medical device excise tax set to go into effect in 2013 during the first Jobs & Innovation Forum this week.
Rodgers called the FDA’s device review process "onerous and inconsistent," calling for fewer unnecessary regulations on med-tech to spur innovation and job-creation.
St. Jude’s lands two industry firsts with regulatory wins for chronic migraine neurostim treatment and quadripolar CRT win in India | Regulatory Roundup

St. Jude Medical Inc. (NYSE:STJ) announced a med-tech first with a European CE Mark approval for its Genesis implanted neurostimulation device for patients with chronic migraines.
Genesis, which is the first device of its kind to win regulatory approval in any market, provides peripheral nerve stimulation to the occipital nerves to manage migraines. Small electrical leads are placed under the skin and connected to a device which produces pulses of stimulation.
Cancer therapy device maker Delcath drops hard on inconclusive trial results
Delcath Systems Inc. (NSDQ:DCTH) saw share prices drop 11 percent on news that its flagship cancer therapy device presented inconclusive results in treatment of colorectal cancer.
Sixteen patients with very late stage colorectal cancer participated in Delcath’s phase II study of its Chemosat focused chemotherapy delivery system, which uses a series of tubes to aim chemotherapy drugs at a specific organ and then filter the blood to remove the toxins before returning it to the body.


