
Edwards Lifesciences (NYSE:EW) dipped below $70 per share, delighting one prominent Wall Street watcher but failing to thrill analysts at Piper Jaffray.
The Medical Device Business Journal — Medical Device News & Articles | MassDevice

Edwards Lifesciences (NYSE:EW) dipped below $70 per share, delighting one prominent Wall Street watcher but failing to thrill analysts at Piper Jaffray.
Comprehensive Settlement Covers Palomar’s Lawsuits Against Candela and Syneron
SCOTTSDALE, Ariz. and HAYWARD, Calif., Sept. 13, 2011 — Medicis Pharmaceutical Corporation (Medicis) (NYSE:MRX) and Solta Medical, Inc. (Solta; Solta Medical) (Nasdaq:SLTM) today announced that the two companies have entered into a Stock Purchase Agreement for Solta to acquire all of the outstanding shares of Medicis Technologies Corporation (formerly LipoSonix, Inc.), a subsidiary of Medicis.
Baxter International Inc. (NYSE: BAX) won an expanded indication for its Artiss fibrin sealant, making it available for use during in sealing tissue flaps during face-lift surgeries.
Artiss first won FDA clearance in 2008 for use in adhering skin grafts in wound beds prepared for treating burns in adults and children over the age of one.

GI Dynamics Inc.’s EndoBarrier gastrointestinal sleeve won Australian approval for treatment of type 2 diabetes and obesity.
The Lexington, Mass.-based company’s device is a plastic sleeve inserted endoscopically into the small intestine, where it slows the uptake of nutrients from food to induce weight loss and help control the symptoms of diabetes. The TGA approved EndoBarrier for up to 12 months of use.

Medicis Aesthetics Inc. is asking the FDA to expand the use of its flagship Restylane facial wrinkle filler for lip augmentation procedures.
The Scottsdale, Ariz.-based division of Medicis Pharmaceutical Corp. (NYSE:MRX) secured a panel date with the FDA’s General and Plastic Surgery Devices Panel April 27.

Syneron Medical Ltd. (MKT:ELOS) consolidated its consumer business into a new spinout called Syneron Beauty.
The subsidiary of the Yokneam, Israel aesthetic device company will conduct the sales, marketing and distribution of all of Syneron’s consumer products. That includes Syneron’s home-use partnership with Procter & Gamble, the Tända light therapy products, the Mē hair removal system and the Fluorinex teeth whitening system.
The new unit will be led by Fabian Tenenbaum, who previously served as Syneron’s CFO.
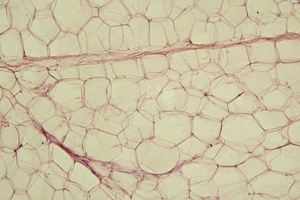
A patient’s own cells may be able to create fat tissue with its own blood supply.
Image courtesy Jagiellonian University Medical College

In an apparent fire sale, Elemé Medical Inc. has sold off all its assets to larger competitor Cynosure Inc. (NSDQ:CYNO) for just less than half of its annual sales.
Elemé, which manufactures the SmoothShapes laser cellulite removal system, was generating about $5 million in annual sales and had raised more than $25 million from investors since 2008.

Shares of Allergan Inc. (NYSE:AGN) stock were down nearly 2 percent after a Food & Drug Administration review found that women with breast implants have a greater chance of contracting a rare form of cancer.
The federal watchdog agency warned doctors and patients of a possible link between saline- and silicone gel-filled breast implants and anaplastic large-cell lymphoma, a very rare type of cancer.

