There doesn’t seem to be much stopping the obesity epidemic in the U.S., with ⅓ of the population considered dangerously overweight – but not for lack of trying, as evidenced by a glut of diets, pills, programs and surgeries aimed at helping people lose weight.
Cutting through the clutter and helping patients get lasting results is something ReShape Medical is hoping its Duo system can accomplish. Richard Thompson, CEO of the San Clemente, Calif.-based medical device maker, told MassDevice.com that the Duo system combines a relatively non-invasive surgical procedure with a comprehensive program that teaches patients how to keep the weight off long after the treatment is completed. That combination, Thompson told us, is necessary to help the obese retrain their bodies and serves as an alternative to more invasive weight loss surgeries.
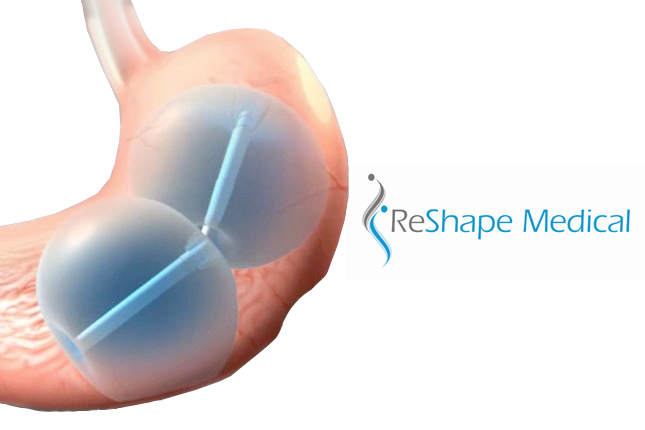
The ReShape Duo is a dual balloon system designed to be inserted endoscopically and remain in the stomach for 6 months. The balloon takes up space in the stomach and helps patients feel full. While the stomach-filling balloons are in place, patients are counseled by healthcare professionals on nutrition, exercise and behavior modification to help them develop a healthier lifestyle, according to ReShape.
The company, which filed for pre-market approval from the FDA in the summer of 2014, recently released results from its Reduce US pivotal trial that it said showed patients with the device lost 2.3 times more weight than patients using just diet and exercise.
The Duo, which has been available comercially in Europe since 2007, is a non-reimbursed cash procedure, which Thompson said will continue when it hits the States, hopefully sometime this year. That’s good news for ReShape, which has raised north of $40 million since its inception.
MassDevice.com sat down with Thompson recently to discuss ReShape’s progress and why past attempts to fight the obesity epidemic have failed.
MassDevice.com: Can you describe the ReShape Duo procedure for us?
Richard Thompson: Intragastric balloons have been around for quite a while in Europe and in other countries, but not in the United States. The original designs were just simple, spherical balloons. They’re very, very simple devices. You put a balloon in somebody’s stomach and it takes up space. What our team did is they looked at it and said, "That’s a great start, but what would the next generation product look like?"
They determined that you could come up with a design that fills the stomach more fully than you can do with a single sphere, causing patients to eat even smaller meals and theoretically lose even more weight. Our product is called the ReShape Duo because it has 2 balloons. Each of our balloons is slightly smaller than the original product’s single balloon. Together they represent almost twice as much volume in the patient’s stomach, so patients will eat even less. We’ll see over time how much more weight they actually lose.
The 2nd thing is those 2 balloons are connected with a flexible shaft. Because the stomach is C-shaped, those 2 small balloons conform to the stomach’s shape. They’ll nestle down into the stomach and curve with the stomach for better comfort and better stability of the device.
MassDevice.com: Why is it the right time for a device like the ReShape Duo?
Richard Thompson: The unmet need is not new. For quite a long time, patients in the United States have had limited options for helping them treat their obesity. They’ve always been able to try diet and exercise, but it’s well known that often [diet and excercise] doesn’t allow them to meet their needs and meet their objectives. Then they could also go completely to the other end of the spectrum and choose a surgical option, which is definitely going to cause them to lose weight but with consequences and with a very severe procedure.
In between those 2, there has been an unmet need that no one’s ever been able to get a product on to the market in the U.S. to meet. I will say that the reason that this product is going to be coming on the market, as we expect it to be coming on the market soon, is really because of a shift at FDA.
Over the last 3 years, the FDA has made it clear that they’ve moved to a more favorable approach to bariatric devices and ReShape was fortunate enough to be early in line to get a pivotal trial done through FDA. That’s probably what determines the timing of this product coming to market more than anything else.
MassDevice.com: Looking at the landscape, including the surgical interventions you talked about, plus diet, apps, fitness programs, it seems that the only common denominator is that they’re not making a dent in the obesity epidemic. Take me through some of the difficulties in tackling this problem.
Richard Thompson: First on the surgical end of things, about 1% of the patients who are qualified for a surgical procedure actually choose to have those procedures, and the reason is pretty straightforward. They just don’t like the idea of having their internal organs re-arranged on a permanent basis, the consequences that come with that and the lifestyle changes that have to be made to accommodate that. Plus a lot of patients see that as failure. They feel they want to solve their obesity problem, but they want to solve it to themselves. They don’t want somebody to change them in order to solve it. That’s why on that end of the spectrum you don’t see a big dent in this market.
On the diet side, you certainly see a lot of attempts. Almost everybody who’s overweight will try the various diet programs and exercise programs to get their weight under control, but it requires a tremendous will ower and compliance to stick with it.
Almost everybody can lose some weight when you do a diet and do an exercise program, but the willpower and stick-to-itiveness that’s required is beyond what most people can actually sustain, so the weight comes back. What makes our product different is that when you put a balloon in a patient’s stomach, it is a physical change but it’s not a permanent change. Now the patient’s stomach becomes full after eating a very small meal and the patient feels full after a very small meal has been consumed, but that small meal means fewer calories. Therefore, they’re going to lose weight.
What’s particularly different about our type of therapy is there’s a feedback mechanism going on here. The patient eats a small meal, feels full, loses weight. The next day they eat a small meal, they feel full, they lose weight. Using behavior modification therapy and diet and exercise counseling you can really cement that relationship, so that the patient gets used to this idea that, "Yeah, guess what? If I eat small meals, I will lose weight. After the balloon is out, if I continue to eat small meals like I’ve been doing for the last 6 months when the balloon is in, I can keep that weight off."
I think it’s the power of that feedback mechanism that really gives our particular therapy a major advantage over some of the other device therapies that are under development.
MassDevice.com: Is this ReShape device an alternative to a surgical procedure such as the Lap-Band?
Richard Thompson: Not really. Patients who want to have those procedures, are willing to do that, will probably do it. Our product is going to appeal to the patients who will never consider a surgical procedure, which is more than 99% of patients. We’re not going to be competing with Lap-Band. We’re not trying to persuade people that want a Lap-Band that they shouldn’t have it. We’re trying to provide an alternative for people who have nothing.
MassDevice.com: How much of your job is getting patients to understand that there’s no magic bullet or cure? How do you reach people who are chronically overweight or obese but still say, "If I just do this diet or if I just do the procedure, then I’ll be all set and done?"
Richard Thompson: Those are the wrong patients for our procedure. We don’t want people who need and want a magic bullet to buy this product, because they won’t be happy with it. It does take a lifestyle change. They have to put the hard work in to actually reap the full benefit of the product.
Everybody will lose weight if you put the fork down, just about everybody. When you check it out, that’s where the rubber meets the road and that’s when you need the motivated patient. What we see when the product is used commercially in Europe is that the patients self-select to be motivated. They have to pay roughly $8,000 or €5,000 for this procedure in Europe. They’re already saying, "Okay, I am so motivated I am going to put that much money on the line."
They enter into this arrangement then with the doctor who’s going to manage them through this procedure and the doctor, instead of running a clinical trial where he’s measured by how well did he run a clinical trial, the doctor is now motivated to maximize this patient’s experience and make them a happy person at the end of the procedure.
MassDevice.com: Let’s dig back into the pricing in terms of the counseling afterwards. You’re saying a lot of these bariatric surgeries already have built in dietary lifestyle and things like that? Do you imagine the patient will pay one lump sum and then get a year’s worth of service?
Richard Thompson: Yes.
MassDevice.com: How economically does that work for you guys? Are you partnering with them to help provide the education or are you just providing the device?
Richard Thompson: We’re providing the device. We’re giving them the template for how to do the counseling. We’re going to be providing things like web tools, a web portal for the patients that the physicians will also use. We’ll be providing them with services and tools that they can use. It will really be part of the price that the patient pays to the bariatric surgeon will cover all of the counseling that they will get.
MassDevice.com: So you’re not building a specific dietary program?
Richard Thompson: We’ll have the template. We’ll have program laid out for how we think it should be done. There’s going to be some customization of that, every dietician’s got their own ideas about exactly how to do it. What’s important to us is that we’re well-aligned in terms of what we’re trying to accomplish with the patient. We want the patient at the end of the day to meet their weight lose objectives.
As long as they’re dedicated to that and they have the willingness and ability to implement a meaningful program for the patient, I think we’ll be happy with that to the extent we can get them to comply with ours the better because then we can have a uniform experience across the country. I’m sure there will be some variability in there.
MassDevice.com: Where is the procedure performed?
Richard Thompson: Outpatient surgery centers, there’s quite a few bariatric surgery centers that are not based on a hospital. That will be the starting place.
MassDevice.com: Will you work with bariatric surgeons in terms of helping them build their practices?
Richard Thompson: We definitely will be helping them with that, because we’ve talked about this behavior modification patient counseling being such an important part of the program we’re offering. The primary reason we’re going to bariatric surgeons in the first place is because they offer those programs to their surgical patients. They already have the infrastructure and personnel and skillsets that we need to use to implement in our program, though ours will be a little different than what they do for surgical patients. They have the basic endoscopy skills that we need to have.
MassDevice.com: Yeah. What other marketing channels do you anticipate using? For example, direct-to-consumer?
Richard Thompson: When we go out with this product initially, we’re not just going to be going major television and tap the product. We don’t want to go out too fast. We need to learn some things. We need to learn the best way to implement this program, so we’ll go to a relatively small number of sites, work with them for a period of time and then expand. Figuring out exactly what we need to learn, where we’ve made mistakes, where do we need to make changes? That’s probably our biggest challenge.
MassDevice.com: You just filed your PMA application this summer. How has the interaction with the FDA been?
Richard Thompson: We have a very positive, collegial relationship with FDA. Those are terms you don’t hear very often with FDA, but in this case, we get good feedback from them and everything we’re hearing is positive. It doesn’t tell us very much about timing, but if the timing is normal then we should have the FDA’s decision some time in the 1st half of this year.
MassDevice.com: Obviously there’s huge upside potential, but also some big challenges. What keeps you up at night?
Richard Thompson: Frankly I’m not terribly worried about the marketing of the product set. The commercialization of this product I think is going to go well. We will definitely learn and modify as we go, but if you look at the history of the bariatric states, it’s filled with failure. That makes funding of startup companies like we are difficult, because you can’t point to a company and say, "Well, we’re like that right there. Why don’t you give us some money and we’ll do what they did?" We actually have to be 1st and be the 1 that paves the way for everybody else. That’s our biggest challenge, getting the right investors involved and getting the company well-funded.
The greatest concern about using intragastric balloons from a safety point of view is that if the balloon should deflate prematurely, it has the potential to migrate into the intestines where it can cause an obstruction. That doesn’t happen a lot. When it does happen, it’s a very serious emergency event and that requires surgery to go in and get it out. With 2 balloons, the balloons are independent of each other even though they’re connected. They’re fluidically independent of each other. If 1 of the balloons goes down, the other balloon remains inflated and acts as an anchor to keep the entire device in the stomach. These balloons are filled with saline that has been colored dark blue by a dye that we put in there and when that dye is released from a deflated balloon, it goes into the patient’s urine and it turns the patient’s urine very dark blue or dark green. The patient is trained to recognize that as a signal that, "Something has happened here. I need to go see the doctor." The doctor can then go in and retrieve that balloon because it’s been retained in the stomach. It’s very routine task to do that in those surgery involved.