
Monarch Medical’s mission is to transform how hospitals and caregivers handle "drugs of concern," or compounds that, when mismanaged, can lead to injuries or even death – the aptly named "never events" that ought to be anathema to healthcare.
The Medical Device Business Journal — Medical Device News & Articles | MassDevice

Monarch Medical’s mission is to transform how hospitals and caregivers handle "drugs of concern," or compounds that, when mismanaged, can lead to injuries or even death – the aptly named "never events" that ought to be anathema to healthcare.


By Sony Salzman
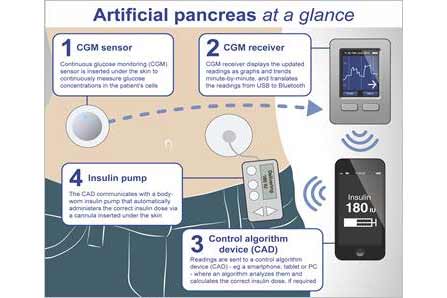
Two medical technology partnerships are racing to develop artificial pancreas technology that might represent a new chapter in Type I diabetes management.
An artificial pancreas would automate communication between portable, personal glucose monitors and insulin pumps to help manage diabetes without medical supervision.
By Tripp Underwood
For decades, patients have managed their type 1 diabetes by injecting themselves with insulin to regulate the glucose in their blood. While this form of medical management addresses the immediate danger of low insulin levels, long-term complications associated with diabetes, like heart and kidney diseases, still threaten more than 215,000 children currently living with the disease in the United States.

Diabetes devices maker Valeritas announced today that it closed a structured debt financing with $100 million to support commercial efforts of its V-Go insulin delivery system.
The new funding adds to Valeritas’ previously raised $150 million in equity, as well as continued support from the company’s private equity and venture capital backers, the company said.

MASSDEVICE ON CALL — Healthcare regulators down under issues a safety advisory for a trio of potential problems with certain Medtronic’s (NYSE:MDT) Paradigm insulin pumps.
The Australian Therapeutics Goods Admin. warned patients of 3 issues, but noted that "these issues are rare and it is unlikely that you will need to replace your Medtronic Paradigm Insulin Pump."

The FDA put its highest-risk category on Johnson & Johnson (NYSE:JNJ) subsidiary Animas Corp.’s insulin pump recall over concerns that the devices may cause patient harm or even death.


MASSDEVICE ON CALL — Researchers touted positive outcomes for the 1st study to compare a traditional insulin pump to a dual-hormone "artificial pancreas" in treating patients with diabetes.
The Canadian team reported that patients using the experimental technology showed improved glucose levels and lower risks of hypoglycemia, potentially advancing research into a commercially viable artificial pancreas.

