InSightec said today that it reeled in a $59 million Series D round from institutional investors in the U.S. and Asia and named Dr. Maurice Ferré as chairman of the board.
InSightec developed the ExAblate MR-guided ultrasound targeting technology.
The Medical Device Business Journal — Medical Device News & Articles | MassDevice
InSightec said today that it reeled in a $59 million Series D round from institutional investors in the U.S. and Asia and named Dr. Maurice Ferré as chairman of the board.
InSightec developed the ExAblate MR-guided ultrasound targeting technology.



Israeli device maker InSightec added $12.5 million to its coffers thanks to the support of Chinese backers, the firm’s parent company announced.

Israel-based InSightec is looking for a few investors to jump in on its $50 million Series D funding round, saying it’ll use the funds to boost commercial efforts for its non-invasive tumor-targeting ultrasound therapy.
York Capital Management is already lined up to take a chunk out of the $50 million, leaving "limited time" for other prospective funders to jump in on the initial round or to expand fundraising to $62.5 million, InSightec said.
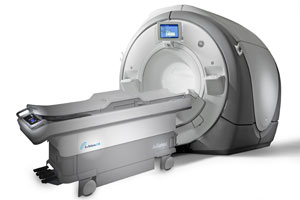
Canadian healthcare regulators approved InSightec‘s ExAblate ultrasound system, clearing the device for use against 2 different diseases. Health Canada granted ExAblate indication to break up benign uterine tumors and to alleviate painful symptoms of metastatic bone tumors.

Israeli device maker InSightec landed a green light from the Chinese Food & Drug Administration for its ExAblate MRI-guided focused ultrasound therapy, with an indication to treat non-cancer-causing tumors in the uterus.
These tumors, called uterine fibroids, occur in more than 20% of women of childbearing age, according to the company. ExAblate is an incision-free treatment that uses ultrasound frequencies to ablate tumors.

InSightec picked off $30.9 million in a Series C financing round, led by a $27.6 million investment from GE Healthcare and $3.3 million by other investors, the company said.
In addition, the Israeli-based ultrasound maker also named VP & general manager of the MRI business James Davis as new CEO and Tom Gentile as chairman of board. Gentile most recently was
president & CEO of GE Healthcare.

Israeli device maker InSightec won FDA approval for its ExAblate MRI-guided focused ultrasound therapy with indication to treat pain resulting from bone metastases in patients who failed or aren’t good candidates for radiation therapy.
Bone metastases occur when cancer cells break off from the primary tumor site and spread to other parts of the body, commonly leaving patients with significant physical and emotional pain, according to a press release.

The FDA approved U.S. studies of Israel-based InSightec’s ExAblate Neuro in treatment of medication resistant Parkinson’s disease.
InSightec will enroll 30 patients who suffer from tremors associated with the disease, randomizing them for treatment with ultrasound-based deep-lesioning of the brain.

