EuroPCR
EuroPCR 2014: Looking for hope for renal denervation

Amid the hand-wringing and back-pedaling surrounding renal denervation, some physicians are still looking for ways to use the technology to treat patients with hypertension.
EuroPCR 2014: St. Jude touts EnligHTN data amid renal denervation backlash

EuroPCR 2014: Docs say renal denervation ads are putting ‘marketing before science’

Attendees at this year’s EuroPCR conference in Paris cast a wary eye on medical device makers’ efforts to promote renal denervation in treatment of hypertension, taking to Twitter to note and sometimes chide companies for putting "marketing before science."
EuroPCR: Volcano touts positive early results for its lesion assessment system

Volcano Corporation (NSDQ:VOLC) posted positive early findings for its instant wave-Free Ratio (iFR) coronary lesion detector, with results form its Advise II trial supporting results from older retrospective publications, according to a report unveiled during a hot line late-breaking clinical trial session at the EuroPCR meeting in Paris, France.
EuroPCR: 1-year results continue to support Edwards’ next-gen Sapien XT valve
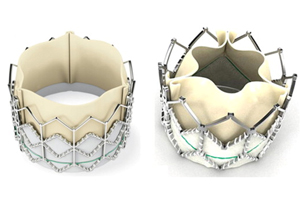
Edwards Lifesciences’ original Sapien (left) and new Sapien XT (right) valves.
EuroPCR: Boston Scientific’s Lotus TAVI bests rivals with less leakage

Massachusetts-based medical device titan Boston Scientific (NYSE:BSX) released early data from a study of its Lotus transcatheter aortic valve implantation system, highlighting very low rates of paravalvular leakage that analysts said could give the company some leverage in the highly charged TAVI market.
MassDevice.com +7 | The top 7 med-tech stories for the week of May 14, 2012.

Say hello to MassDevice +7, a bite-sized view of the top seven med-tech stories of the week. This latest feature of MassDevice.com’s coverage highlights our seven biggest and most influential stories from the week’s news to make sure you’re up to date on the headlines that continue to shape the medical device industry.
If you read nothing else this weekend, make sure you’re still in the know with MassDevice +7.
EuroPCR: Renal denervation devices take center stage
CardioKinetix’s cath-based Parachute may “revolutionize” treatment
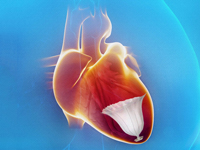
CardioKinetix made strides in research for its novel Parachute catheter-based left ventricle implant, which proved promising after 2 years in study results unveiled at this year’s EuroPCR conference in Paris.
The Parachute is inserted into the heart via a catheter threaded through the femoral artery. Once inside the heart, the device springs open and attaches to the walls of the heart to provide support for patients whose left ventricle was left enlarged after a heart attack.
EuroPCR: “Excellent” safety, effectiveness for BSX’s Promus Element in small vessels

Boston Scientific (NYSE:BSX) unveiled new study results at the EuroPCR meeting in Paris today, touting the "excellent" safety and effectiveness of its Promus Element drug-eluting stents in small coronary vessels.



