(Reuters) — Bayer (ETR:BAYN) has short-listed India’s Lupin (NSE:LUPIN) and investor BC Partners as prospective buyers of medical skin cream assets in a potentially €1 billion ($1.1 billion) deal, several people familiar with the sale told Reuters. Germany’s Bayer has for more than 2 years sought to narrow the focus of its healthcare division and its planned $66 billion Monsanto […]
Dermatology
Sensus Healthcare postpones IPO
Sensus Healthcare reportedly postponed its initial public offering today, after setting terms earlier this month with a midpoint raise of $20 million. Boca Raton, Fla.-based Sensus makes the SRT-100 and SRT-100 Vision photon X-ray low-energy superficial radiotherapy systems for treating non-melanoma skin cancers, including basal cell and squamous cell carcinomas. The company said March 9 that it planned […]
Mela Sciences recalls MelaFind
Mela Sciences (NSDQ:MELA) recalled its flagship MelaFind melanoma diagnosis aid because some of its software wasn’t included in its pre-market approval from the FDA.
Smith & Nephew gets in on Michelson Diagnostics Series B round
Derma Sciences skins out $36M

Derma Sciences (NSDQ:DSCI) said it’s closed an offering of some 3.5 million shares of its stock at $10.34 apiece.
The Princeton, N.J.-based medical device company said it plans to use the $33.8 million in net proceeds from the $36.4 million offering to further develop its lead drug candidate DSC127.
Derma Sciences said the drug is being designed to treat chronic diabetic foot ulcers.
MassDevice.com +3 | The top 3 med-tech stories for March 22, 2012.

Say hello to MassDevice +3, a bite-sized view of the top three med-tech stories of the day. This feature of MassDevice.com’s coverage highlights our 3 biggest and most influential stories from the day’s news to make sure you’re up to date on the headlines that continue to shape the medical device industry.
If you read nothing else today, make sure you’re still in the know with MassDevice +3.
Mela Sciences’ Joe Gulfo on returning to the market in the shadow of the medical device tax

Mela Sciences (NSDQ:MELA) CEO Joe Gulfo tells MassDevice that he feels like a kid in a candy store as he takes the company back to market – and not even the ever-looming burden of the medical device tax can dampen his enthusiasm.
SBi secures $43M loan | Funding Roundup
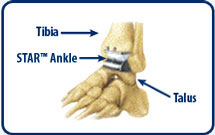 Star total ankle replacement
Star total ankle replacementOrthopedic technology company Small Bone Innovations Inc. landed a $43 million loan to further sales of its flagship ankle replacement device.
The New York-based company plans to use the credit facility as working capital to drive U.S. growth of its Star total ankle replacement, a non-cemented implant designed to replace arthritic ankle joints in patients with osteoarthritis, post-traumatic arthritis or rheumatoid arthritis.
The system is currently the only mobile-bearing total ankle replacement approved for the U.S. market.
What’s up with Mela Sciences? | Wall Street Beat

If the FDA grants clearance and no-one hears, did it really happen?
That might be the question of the day for Mela Sciences (NSDQ:MELA) and CEO Dr. Joseph Gulfo, as they try to comprehend a 15.4 percent share price plunge, despite a long-awaited approval granted by the federal watchdog agency just a few days ago.
Mela Sciences CEO: MelaFind rollout planned for early 2012

Dr. Joseph Gulfo, CEO of Mela Sciences (NSDQ:MELA), says the company plans to begin rolling out its MelaFind skin cancer detection system during the first quarter of 2012, after winning a hard-earned green-light from the FDA.
"I don’t think we’ll be ‘selling,’ in the classic sense of selling, for at least a year and a half," Gulfo told MassDevice. "Dermatologists want this system, so we don’t need to ‘sell’ it.
Mela shares double overnight after positive FDA review

Mela Sciences Inc.’s (NSDQ:MELA) stock doubled in price overnight after the a Food & Drug Administration panel voted in favor of its melanoma-detecting device’s safety and efficacy.
MELA shares were up 105.5 percent to $5.24 before the trading day opened. Through yesterday’s close, the shares lost 60 percent of their value in the past three months. The company’s shares’ gain remained around 95 percent up in mid-morning trading.



