
Top 3 reasons your 510(k) submission was rejected – Part I

The Medical Device Business Journal — Medical Device News & Articles | MassDevice


By Mark Healey

By Ames Gross, Pacific Bridge Medical
On December 17, 2013, the China Food and Drug Administration (CFDA) announced a streamlined device registration renewal process that took effect on January 1.

by Marshall Allen, ProPublica

By Peter M. DeComo, chairman and CEO of ALung Technologies, Inc.
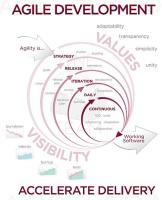
By: Rod Cain

The Massachusetts Medical Device Development Center at the University of Massachusetts Lowell (M2D2) is pleased to announce and host the M2D2 New Venture Competition 2014 to showcase the innovative developments of early-stage medical device companies. Companies will compete for $50,000 of services from M2D2, Nutter McClennen & Fish LLP, and Smith & Nephew, Inc.
For full contact details please see the Call for Application and Entry Form Below.

Nancy Singer for Sparta Systems

By John Baker
Through our experience, we have found the biggest challenge in recruiting and placing senior level Regulatory Affairs and Quality Assurance Executives, is finding the right balance of technical knowledge, communication skills and leadership qualities.

(Dave Herholz, Flickr)
by Michael Grabell, ProPublica

