Acutus Medical said today that it agreed to acquire Rhythm Xience for an undisclosed amount and inked deals with a trio of new partners. Eden Prairie, Minn.-based Rhythm Xience makes a line of transseptal crossing and steerable introducer systems including the Flextra, Guider and Lancer devices, which are all 510(k)-cleared by the FDA. “At Acutus, we […]
Cardiac Designs
FDA warns iPhone ECG maker Cardiac Designs
The FDA published a warning letter it sent to Cardiac Designs over issues associated with the company’s lack of design validation and how it handles and analyzes complaints about its device. The letter was sent after an FDA inspector visited the company’s Round Rock, Texas location in June. Utah-based Cardiac Designs produces an Apple (NSDQ:AAPL) iPhone-based electrocardiogram […]
FDA clears new smartphone health monitoring technologies

A growing demand for mobile health monitoring devices led to a boom in mobile devices that allow patients to manage chronic illnesses and measure body metrics at home. Now, several companies have won FDA clearance for next-generation health monitoring devices that connect wirelessly to mobile apps and software.
Palo Alto, Calif.-based Glooko will begin selling its second glucose monitoring app and cable device. This product comes with enhanced software that allows patients to program health goals and enhanced data security through advanced encryption.
AliveCor revamps mobile ECG for iPhone 5, hands out free samples

California startup AliveCor updated its heart monitor to make it compatible with the next-generation iPhone 5. The company’s mobile ECG snaps on to the outside of an iPhone with sensors that monitor and record electrocardiogram rhythms wirelessly.
FDA clears over-the-counter iPhone ECG
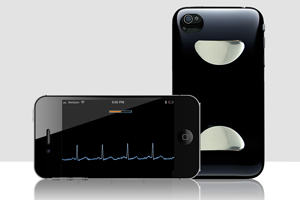
Utah-based Cardiac Designs is still flying under the radar after having won FDA 510(k) clearance for its iPhone-based ECG monitor.
The FDA nod may represent a win for consumers who are increasingly interested in recording their own health data, but the regulatory win comes with a strange restriction – patients must obtain a physician prescription in order to view the raw wave form data captured from the heart.

