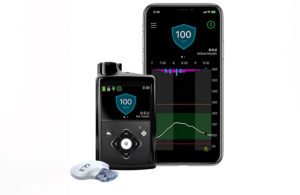
Results from the ADAPT study, published in The Lancet Diabetes & Endocrinology, demonstrated improved glycemic control and treatment satisfaction among users of MiniMed 780G.
The MiniMed 780G system is available in more than 60 countries. It is currently under FDA review. Medtronic is hopeful for clearance during the current fiscal year. If clearance takes place, it would be good news for a Diabetes business that has faced regulatory troubles. (The company recently said that it’s achieved 90% of the FDA’s action items.)
Get the full story at our sister site, Drug Delivery Business News.

