The FDA upgraded personal massage device maker King International LLC’s voluntary recall of its ShoulderFlex system to a Class I designation, the most serious category, after one patient was killed, one was strangled and two others were injured by the device.
The federal watchdog agency’s Class I designation is applied to devices where there is a reasonable probability that the products will cause series health consequences or death.
The ShoulderFlex recall applies to nearly all of the 12,000 massagers on the market in the U.S., which were sold between 2003 and 2011 through stores, catalogs and online retailers.
Sign up to get our free newsletters delivered straight to your inbox
The federal watchdog agency issued a warning about the ShoulderFlex system in August asking health care providers not to recommend the device and urging consumers to dispose of the pieces separately so that they can’t be reassembled and used.
"The FDA is concerned that the ShoulderFlex Massager presents serious health risks," according to the warning. "Hair, clothing, and jewelry can become entangled in the massage device and cause serious injury or death."
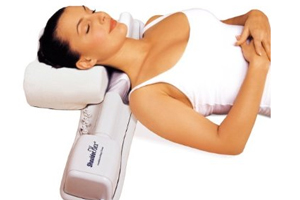
ShoulderFlex is a therapeutic massager that provides deep tissue massage to the neck, shoulder and back while a user lies on it. It contains a rotating bar with removable "fingers" that can be adjusted to modify the massage.
The incidents leading to the warning and upcoming recall occurred when a necklace and piece of clothing got caught in the device’s rotating massager. The FDA noted that it had two other reports of hair and clothing being caught in the device.
Click here to view a promotional demonstration of the ShoulderFlex massager. The video advises users to keep hair and jewelry away from the rotating bar.

