Dr. Calvin Ng and Dr. Rainbow Lau performed the first use of Galaxy at Prince of Wales Hospital. The procedure marks the beginning of the first clinical trial of the robotic bronchoscopy system in Asia.
San Carlos, California-based Noah Medical received FDA clearance for the Galaxy system in March. The company then reported the first commercial cases with the system in the U.S. in May.
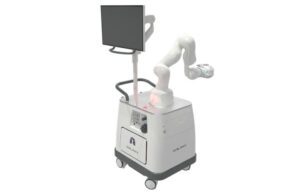
The system provides bronchoscopic visualization and access, enabling diagnostic and therapeutic procedures in patient airways. Galaxy features advanced imaging technologies that provide real-time location updates for potentially cancerous lesions. Noah Medical designed the technology to improve tool-in-lesion and diagnostic yield. Physicians can use it to confidently navigate and biopsy lung nodules.
“We are excited to be the first site in Asia to trial this exciting new technology,” said Ng. “The Galaxy System is the latest generation in robotic bronchoscopy and we are very much looking forward to seeing this being used in Hong Kong, and eventually the broader Asia region.”
Galaxy features proprietary integrated tomosynthesis (TiLT+ Technology) with augmented fluoroscopy. It has a disposable, single-use bronchoscope and a small, compact footprint for easy integration into most bronchoscopy suites.
Noah Medical expects the Hong Kong trial to further validate its TiLT+ Technology, center-strike accuracy and overall diagnostic yield. The first patients have been enrolled successfully and the trial is currently underway.
“Congratulations to Dr. Ng, Dr. Lau and the team at Prince of Wales Hospital for their continued efforts in bringing advanced robotic lung technologies to Asia,” said Jian Zhang, Noah Medical founder and CEO. “We are excited to explore the opportunity of early diagnosis and treatment of lung cancer globally, which will benefit billions of patients.
“Our mission, in concert with pioneers like Dr. Ng and Dr. Lau, is to help physicians diagnose and treat lung cancer more accurately and efficiently than ever before.”

