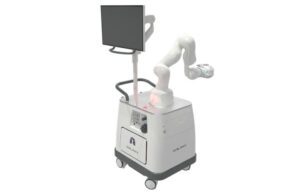
San Carlos, California-based Noah Medical designed Galaxy and its accessories to provide bronchoscopic visualization and access. These capabilities provide diagnostic and therapeutic procedures in patient airways. The system features advanced imaging technologies that provide real-time location updates for potentially cancerous lesions. Noah designed the technology to improve tool-in-lesion and diagnostic yield.
Galaxy uses proprietary integrated tomosynthesis, called TiLT Technology. It enables augmented fluoroscopy with a disposable single-use bronchoscope with always-on vision, and a small, compact footprint that allows for easy integration into most bronchoscopy suites.
Dr. Krish Bhadra of CHI Memorial in Chattanooga, Tennessee, performed the milestone procedure. CHI is also the first hospital in the state to adopt Galaxy, marking another step for Noah in the robot’s commercial rollout. Galaxy first received FDA clearance in March 2023, with its first commercial use in the U.S. coming in May.
In a news release, Bhadra compared lung biopsies with current tools and first-generation robotics to “flying an airplane blindfolded.” The surgeon said Galaxy “fundamentally changes the game,” possibly reshaping the treatment of cancerous lesions.
“Congratulations to Dr. Bhadra and the team at CHI Memorial for adopting and advancing image-integrated robotic assisted bronchoscopy,” said Jian Zhang, Noah Medical founder and CEO. “Our journey to 500 successful cases in the U.S. using the Galaxy System is just the beginning. We are improving patient care, transforming clinical workflow, and contributing to the future of healthcare.”

