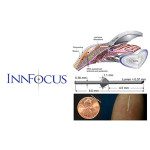
 InnFocus said it closed a $33.9 million Series C round for the upcoming clinical trial of its MicroShunt device for treating glaucoma.
InnFocus said it closed a $33.9 million Series C round for the upcoming clinical trial of its MicroShunt device for treating glaucoma.
Last month InnFocus won the FDA’s approval to add an additional 412 patients to its clinical trial ahead of a bid for pre-market approval. The last phase of the study is slated to begin in January 2016, the Miami-based company said.
When the federal safety watchdog agreed to allow the trial expansion, CEO Russ Trenary said InnFocus is “poised to become the clear leader in treating all stages of primary open angle glaucoma.”
In April, InnFocus said it reached its goal of enrolling 75 patients in a Phase I clinical study for the MicroShunt device.
MicroShunt received CE Mark clearance in January 2012. InnFocus is seeking to have the product approved in the U.S. for the treatment of primary open-angle glaucoma. The company has also been testing the product in conjunction with cataract surgery.

