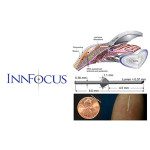
 InnFocus said today it won FDA approval to expand a clinical study of its Microshunt glaucoma drainage system to cover trabeculectomy.
InnFocus said today it won FDA approval to expand a clinical study of its Microshunt glaucoma drainage system to cover trabeculectomy.
The Microshunt system is designed to help reduce pressure within the eye in patients with moderate and late stage open angle glaucoma, and is intended to be an alternative to traditional glaucoma surgery, the Miami, Fla.-based company said.
“As Enrollment for the final phase begins at sites in the U.S. and Europe, and with our recent Series C funding of $32.8 million, the company is poised to become the clear leader in treating all stages of primary open angle glaucoma,” CEO Russ Trenary said in a prepared statement.
InnFocus said it previously met treatment totals required for its Phase 1 trial and safety data it submitted supported a move to the final phase of testing for the device. The company expects to add an additional 412 patients in the final phase of the trial.
“Our experience in Europe indicates that the InnFocus MicroShunt system has the potential of being a revolutionary product for early intervention in the treatment of glaucoma. We have noted a significant and stable drop in IOP and a large reduction in the need for glaucoma medications,” Isabelle Riss of Bordeaux, France said in a press release.
In April, InnFocus said it reached its goal of enrolling 75 patients in a Phase I clinical study for its MicroShunt glaucoma eye-drainage implant.
MicroShunt received CE Mark clearance in January 2012. InnFocus is seeking to have the product approved in the U.S. for the treatment of Primary Open-Angle Glaucoma. The company has also been testing the product in conjunction with cataract surgery.

