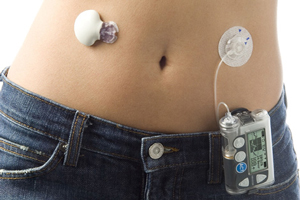
My colleage Gary Schwitzer at HealthNewsReview.org has a post today questioning the validity of observational studies (where epidemiological researchers look at selected variables in large populations to see if there is a relationship between a cause and effect).