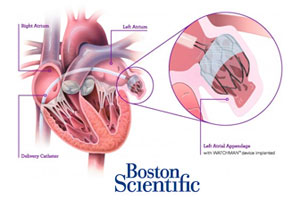
Medical device industry heavy-weight Medtronic (NYSE:MDT) entered a collaboration agreement with ARCA biopharma, providing continuous cardiac monitoring of patients undergoing experimental treatment with a genetically targeted cardiovascular drug therapy.