
Clinical Trials
Recent progress on demographic information and clinical trials
By Barbara D. Buch, M.D.
At FDA, one of our foremost responsibilities is to evaluate and if medical products meets the appropriate standard, to approve or clear drugs, biological products and medical devices. We know that these products are safer and more effective for everyone when they are tested in clinical trials that include diverse populations.
A CDRH Priority: Clinical Trials in the U.S.

Owen Faris, Ph.D., Clinical Trials Director (acting), Office of Device Evaluation in FDA’s Center for Devices and Radiological Health
By Owen Faris, Ph.D., and Jeffrey Shuren, M.D., J.D.
At the Center for Devices and Radiological Health (CDRH), clinical trials are the foundation for our decisions to approve the most important medical devices—products that have the potential to save or sustain life, but that also present the greatest risk to patients.
Clinical trials: Enhancing data quality, encouraging participation and improving transparency
Indian regulators clarify requirements for medical device registrations, clinical trials

By Stewart Eisenhart, Emergo Group
New orders from the Indian Ministry of Health & Family Welfare’s Central Drugs Standard Control Organization (CDSCO) clarify some requirements regarding the country’s sometimes opaque medical device registration and clinical trial regulations.
Filling information gaps for women in medical device clinical trials

By David Strauss, M.D., Ph.D.
At FDA’s Center for Devices and Radiological Health (CDRH), results from clinical trials often serve as the foundation for our decisions to approve the most important medical devices—devices, such as implantable heart devices, that carry the greatest risk to patients and have the potential to save or sustain life.
Abbott closes 3 clinical trials for disappearing drug-eluting coronary stent
Major mismatch: 1 in 3 clinical trials reports inconsistent adverse events, researchers say
A random sampling of clinical trials reported to the federal ClinicalTrials.gov database found that 1 in 3 had inconsistencies in their adverse event outcomes when compared with reports made to peer-reviewed medical journals, researchers said.
Autism clinical trials are ripe for improvement

Medicare denies coverage for Vertos’ spinal device, but leaves room for clinical trials
Updated Jan. 10, 2014, at 4:05 p.m. with comments from Vertos.
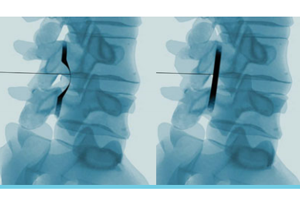
The Centers for Medicare & Medicaid Services this month issued a final decision memo rejecting reimbursement for lumbar decompression in treatment of spinal stenosis, largely reiterating a proposal issued in October.
Neurologic clinical trials in a dish?
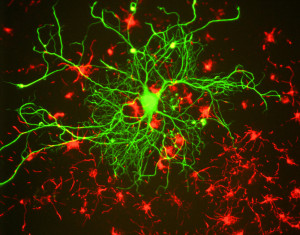
By David Altman




