
Drug-Eluting Stents
Stent Wars: DES best bare metal stents
Updated March 26, 2012 at 9:23 a.m. with new details on Promus/Xience private label deal and March 23, 2012 at 1:15 p.m. with comments from Medtronic.
FDA approves Boston Scientific’s Ion, Taxus stents for heart attack
Boston Scientific’s (NYSE:BSX) Ion and Taxus Liberté drug-eluting stents won pre-market approval from the FDA to treat heart attack patients.
The Natick, Mass.-based med-tech maker said the devices, which elute the anti-restenosis drug paclitaxel, are the only DES approved to treat patients with acute myocardial infarction.
The stents are fashioned from Boston Scientific’s next-generation platinum-chromium alloy. The company said 10% of all coronary interventions are performed for patients having a heart attack.
Federal judge rules for Medtronic, Abbott, Boston Scientific over J&J in DES lawsuit | Legal News

A trio of med-tech titans logged a win against Johnson & Johnson (NYSE:JNJ) in a long-running battle over drug-eluting coronary stent patents.
Federal judge rules for Medtronic, Abbott, Boston Scientific over J&J in DES lawsuit

A trio of med-tech titans logged a win against Johnson & Johnson (NYSE:JNJ) in a long-running battle over drug-eluting coronary stent patents.
Don’t believe the hype: Boston Scientific tackles stent deformation

Boston Scientific (NYSE:BSX), stung by recent studies showing that the thinner design of next-generation stents can deform during deployment, is on the offensive with an new video.
SCAAR: Next-gen DES take the lead from BMS for lowest death risk

Next-generation drug-eluting stents proved safer than their predecessors and their bare-metal cousins for rates of restenosis, stent thrombosis and subsequent death, flipping the tables on previous findings.
New results from Swedish Coronary Angiography & Angioplasty Registry data taken between Nov. 2006 and Oct. 2010 show that DES have finally taken the lead for safety in the stent arena.
St. Jude’s Fame II FFR trial halted on positive results
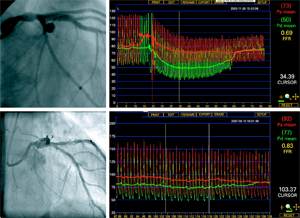
St. Jude Medical (NYSE:STJ) said it’s halting its Fame II trial after an independent safety advisory board recommended the trial be stopped due to strong positive results.
MassDevice.com +3 | Scott Brown ranks high with med-tech makers, another Md. doc charged with over-stenting and FDA adds potential “shrinkage” to BSX’s Promus Element label

Say hello to MassDevice +3, a bite-sized view of the top three med-tech stories of the day. This latest feature of MassDevice.com’s coverage highlights our three biggest and most influential stories from the day’s news to make sure you’re up to date on the headlines that continue to shape the medical device industry.
If you read nothing else today, make sure you’re still in the know with MassDevice +3.
Update: FDA requires BSX to include stent “shrinkage” risk in Promus Element label
Updated December 19, 2011, at 3:45 p.m. with comments from Boston Scientific spokeswoman Denise Kaigler.

Boston Scientific Corp. (NYSE:BSX) must include the potential for stent "shrinkage" in the labeling for its newly cleared Promus Element drug-eluting stent.
FDA requires BSX to include stent “shrinkage” risk in Promus Element label

Boston Scientific Corp. (NYSE:BSX) must include the potential for stent "shrinkage" in the labeling for its newly cleared Promus Element drug-eluting stent.
The decision comes as concerns mount that certain thinner, more flexible next-generation stents may be prone to crumpling after deployment, creating clots in blood vessels that may lead to heart attack.


